In Silico Environmental Risk Assessment: A Computational Revolution in Chemical Safety for Researchers
This article provides a comprehensive overview of in silico environmental risk assessment (ERA), a computational approach that uses mathematical models to predict the environmental fate and effects of chemicals.
In Silico Environmental Risk Assessment: A Computational Revolution in Chemical Safety for Researchers
Abstract
This article provides a comprehensive overview of in silico environmental risk assessment (ERA), a computational approach that uses mathematical models to predict the environmental fate and effects of chemicals. Tailored for researchers, scientists, and drug development professionals, we explore the foundational principles, including tiered approaches and the role of the Adverse Outcome Pathway (AOP) framework. The article details core methodologies like QSAR, read-across, and toxicokinetic-toxicodynamic models, supported by real-world applications in pesticide management and pharmaceutical assessment. We further address current challenges in model optimization and data integration, compare in silico performance against traditional methods, and validate its use through case studies and regulatory acceptance. Finally, the discussion outlines future directions, emphasizing the potential of these tools to enable proactive, animal-free safety evaluation in biomedical research and development.
Defining In Silico ERA: Core Principles and the Shift from Traditional Toxicology
In silico Environmental Risk Assessment (ERA) represents a computational paradigm that uses sophisticated software and mathematical models to predict the environmental fate and effects of chemicals, thereby supporting regulatory decision-making and safety evaluations. Rather than relying solely on live animal testing or physical experiments, this approach leverages the power of computer simulations to analyze molecular structures and predict potential hazards [1]. The core philosophy centers on using non-testing methods to fill data gaps, prioritize chemicals for further testing, and ultimately reduce reliance on traditional animal studies while increasing the efficiency and scope of risk assessments [2] [3] [4].
The development and adoption of in silico ERA have been largely driven by regulatory frameworks such as the European Union's Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), which explicitly advocates for the use of alternative methods to animal testing [4]. This methodological shift is particularly valuable for addressing the challenges posed by the vast number of chemicals in commercial use and the continuous emergence of new substances, including pharmaceuticals, pesticides, and industrial compounds, where traditional testing approaches would be prohibitively costly, time-consuming, and ethically concerning [2] [5].
Core Principles and Methodological Framework
Fundamental Components of In Silico ERA
In silico environmental risk assessment operates through several interconnected methodological approaches, each serving specific functions within a comprehensive assessment strategy:
Quantitative Structure-Activity Relationship (QSAR) Models: These computational models establish mathematical relationships between the chemical structure of compounds (described by molecular descriptors) and their biological activity or physicochemical properties. QSAR models enable the prediction of ecotoxicological endpoints and environmental fate parameters directly from molecular structure, serving as a primary tool for filling data gaps when experimental information is unavailable [1] [6].
Toxicokinetic-Toxicodynamic (TK-TD) Models: These biologically-based models simulate how chemicals are absorbed, distributed, metabolized, and excreted in organisms (toxicokinetics), and how they subsequently exert toxic effects at their target sites (toxicodynamics). They provide mechanistic insights into toxicity processes across different species and exposure scenarios [1].
Physiologically Based Pharmacokinetic (PBPK) Models: PBPK models represent the anatomy, physiology, and biochemistry of organisms to predict the internal dose metrics of chemicals in specific tissues and organs over time. They are particularly valuable for cross-species extrapolation, such as calculating human equivalent doses from animal studies [7].
Dynamic Energy Budget (DEB) Models: DEB models simulate how organisms acquire and utilize energy across various physiological processes, and how chemical stressors alter these energy allocations. They help predict sublethal effects and population-level consequences from individual-level exposure [1].
Read-Across and Chemical Categorization: This technique involves using data from tested chemicals (source compounds) to predict the properties of untested chemicals (target compounds) based on structural and mechanistic similarity. It requires establishing a valid hypothesis for why chemicals can be grouped together [4] [6].
The Tiered Assessment Approach
In silico ERA typically follows a tiered framework that progresses from simple screening-level assessments to more complex and refined evaluations [1]. This stepped approach ensures efficient resource allocation, where higher-tier (and typically more resource-intensive) assessments are reserved for chemicals that warrant further investigation based on lower-tier results.
The diagram below illustrates this conceptual workflow and the interrelationships between different model types within a tiered assessment strategy:
Quantitative Advantages and Applications
Documented Benefits of In Silico Approaches
The adoption of in silico methods in environmental risk assessment provides substantial quantitative advantages over traditional testing paradigms, particularly in resource efficiency and animal welfare.
Table 1: Quantitative Benefits of In Silico ERA
| Benefit Category | Traditional Approach | In Silico Approach | Reference |
|---|---|---|---|
| Cost Requirements | Up to $9,919,000 for conventional pesticide testing | Potential savings of $50-70 billion for assessing 261 compounds | [2] |
| Animal Usage | High (∼8% of experimental animals used for toxicity testing) | Potential reduction of 100,000-150,000 test animals for 261 compounds | [2] |
| Testing Timeline | Chronic toxicity studies can take up to 2 years | Rapid predictions (hours to days) depending on model complexity | [2] |
| Regulatory Efficiency | Limited by data availability for thousands of chemicals | Enables screening and prioritization of large chemical inventories | [4] |
Practical Applications Across Chemical Classes
In silico ERA has demonstrated practical utility across diverse chemical domains and regulatory contexts:
Pesticide Risk Assessment: Computational tools have been developed to predict pesticide exposure in environmental compartments (air, water, soil) and toxicity to aquatic, terrestrial, and soil organisms. Models like AGDISP predict pesticide spray drift, while specialized models like BeeTox distinguish bee-toxic chemicals with high accuracy (specificity: 0.891) [2].
Per- and Polyfluoroalkyl Substances (PFAS) Evaluation: Integrated frameworks combine experimental data with PBPK modeling to calculate human equivalent doses and support the establishment of tolerable daily intakes for problematic compounds like PFOS and PFOA [7].
Pharmaceuticals and Personal Care Products (PPCPs): QSAR tools are employed to screen for persistent, mobile, and toxic (PMT) and persistent, bioaccumulative, and toxic (PBT) properties among emerging contaminants, enabling prioritization of concerning compounds for further testing and regulation [5].
Biofuel Development: In silico assessments evaluate the ecotoxicity of biofuel mixtures based on production conditions, predicting key parameters like biodegradability, bioaccumulation potential, and soil sorption coefficients to optimize for reduced environmental impact [8].
Experimental Protocols and Methodologies
Standardized Workflow for QSAR-Based Hazard Assessment
The application of QSAR models in environmental risk assessment follows a systematic protocol to ensure scientifically defensible results. The methodology described below outlines the process for screening chemicals for PMT/PBT properties, as applied to PPCPs [5]:
Chemical Selection and Representation: Curate a dataset of target chemicals with verified structural identifiers (e.g., Simplified Molecular Input Line Entry System [SMILES] or Chemical Abstract Service [CAS] registry numbers). For the PPCP assessment, 245 substances were included after quality filtering.
Molecular Descriptor Calculation: Generate physicochemical and structural descriptors for each compound using appropriate software tools. These descriptors numerically represent features relevant to chemical behavior and toxicity.
Model Selection and Application: Apply multiple QSAR tools to ensure comprehensive endpoint coverage and model consensus. The recommended toolkit includes:
- OECD QSAR Toolbox (v.4.4 or higher): For mechanistic profiling, category formation, and read-across
- OPERA (v2.9 or higher): For predictions of physicochemical properties and environmental fate parameters
- EPI Suite (v4.1 or higher): For baseline toxicity and environmental persistence predictions
- QSAR-ME Profiler: For additional model verification
Applicability Domain Assessment: For each model prediction, evaluate whether the target chemical falls within the model's applicability domain (the chemical space for which the model was developed and validated). Predictions for chemicals outside this domain should be treated with caution.
Endpoint Prediction and Threshold Comparison: Compare predicted values against regulatory thresholds for persistence (e.g., half-life in water, soil, or sediment), bioaccumulation (e.g., Bioconcentration Factor [BCF]), and toxicity (e.g., Lethal Concentration 50 [LC50]) as defined in regulations such as REACH Annex XIII.
Data Integration and Weight-of-Evidence: Combine predictions across multiple models and endpoints to form a weight-of-evidence conclusion regarding PMT/PBT classification.
Validation and Uncertainty Characterization: Document the reliability of each prediction through measures of goodness-of-fit, predictive performance, and uncertainty quantification.
Integrated In Vitro - In Silico Testing Strategy
Advanced assessment approaches combine high-throughput in vitro bioassays with in silico modeling, as demonstrated in fish ecotoxicology [9]:
In Vitro Bioactivity Testing: Expose relevant cell lines (e.g., RTgill-W1 cells from rainbow trout) to concentration ranges of test chemicals. Measure multiple endpoints including:
- Cell viability (using miniaturized OECD TG 249 assay)
- Cytomorphological changes (using Cell Painting assay)
In Vitro Disposition (IVD) Modeling: Apply computational models that account for sorption effects (binding to plastic labware and cellular components) to predict freely dissolved concentrations that actually interact with biological targets.
Concentration Adjustment: Adjust nominal in vitro effect concentrations (e.g., Phenotype Altering Concentrations [PACs]) using IVD modeling to derive bioavailable concentrations.
In Vitro to In Vivo Extrapolation (IVIVE): Compare adjusted in vitro effect concentrations with in vivo fish toxicity data to establish protective correlations. In validation studies, this approach demonstrated that 73% of adjusted PACs were protective of in vivo toxicity, with 59% within one order of magnitude of measured lethal concentrations [9].
The Scientist's Toolkit: Software and Databases
Successful implementation of in silico ERA requires access to specialized software tools and curated databases. The table below summarizes key resources available to researchers in this field.
Table 2: Essential Computational Tools for In Silico ERA
| Tool Name | Type | Key Features | Application in ERA | Access |
|---|---|---|---|---|
| OECD QSAR Toolbox | Integrated Workflow System | 60+ databases, 150,000+ chemicals, 3M+ data points, read-across capability | Chemical categorization, metabolite prediction, data gap filling | Free |
| VEGA | QSAR Platform | 90+ models for properties, toxicity, and environmental fate | Predictions for bioaccumulation, aquatic toxicity, mutagenicity | Free |
| Toxtree | Rule-Based System | Cramer classification, structural alerts, threshold of toxicological concern | Hazard identification, priority setting | Free, open-source |
| Danish QSAR Database | Predictive Platform | Suite of 30+ models built in Leadscope software | Regulatory screening, early-stage hazard assessment | Free, web-based |
| EPI Suite | Predictive System | Estimates physicochemical properties and environmental fate | Prediction of persistence, bioaccumulation potential | Free |
| OPERA | QSAR Application | Predicts physicochemical properties and environmental fate parameters | High-throughput screening of chemical libraries | Free |
| AGDISP | Exposure Model | Predicts pesticide deposition and spray drift | Assessment of pesticide aerial transport and deposition | Not specified |
Future Perspectives and Challenges
The future development of in silico environmental risk assessment is moving toward more integrated approaches that address current limitations while expanding application domains. Key research priorities include:
Addressing Chemical Mixtures: Developing frameworks to assess the combined effects of multiple chemicals and other environmental stressors, moving beyond single-substance evaluations [1] [2].
Uncertainty Quantification: Improving methods to characterize and communicate uncertainties associated with in silico predictions, particularly for regulatory decision-making [4].
Advanced Model Integration: Creating interoperable computational frameworks that seamlessly combine QSAR, PBPK, TK-TD, and ecosystem-level models [1] [3].
Regulatory Acceptance: Establishing standardized validation protocols and reporting formats (e.g., QSAR Model Reporting Format [QMRF]) to build confidence in computational predictions for regulatory applications [4] [6].
Data Quality and Availability: Expanding curated, high-quality databases for model training and validation, addressing current gaps for specific chemical classes and endpoints [2] [5].
As these challenges are addressed, in silico methods are poised to become increasingly central to environmental risk assessment, enabling more proactive, comprehensive, and efficient evaluation of chemical safety in an increasingly complex chemical landscape.
Tiered Approaches and Weight of Evidence in Problem Formulation
Within the domain of in silico environmental risk assessment (ERA), problem formulation establishes the strategic foundation for the entire evaluation process. It defines the scope, objectives, and depth of the assessment by explicitly considering the specific regulatory or protection goals, the data availability, and the resources and timeframes involved [1]. Tiered approaches and Weight of Evidence (WoE) are two pivotal, interlinked concepts that operationalize problem formulation. A tiered approach provides a stepwise strategy, starting with simple, conservative models and progressing to more complex ones only as needed. Concurrently, a WoE approach offers a structured framework for integrating results from multiple in silico models and, potentially, other data sources to form a robust, reliable conclusion [10] [11]. This guide details the methodologies and applications of these principles for researchers and safety assessors.
Tiered Approaches in ERA: From Screening to Complex Modeling
Tiered approaches are designed to optimize efficiency in risk assessment. They begin with conservative, high-throughput methods to identify substances of potential concern, reserving more resource-intensive, sophisticated analyses for situations where they are truly necessary [1]. The following table summarizes a typical tiered framework for using in silico models in ERA.
Table 1: A Tiered Approach to the Application of In Silico Models in Environmental Risk Assessment
| Tier | Data Situation | Example In Silico Methods | Application Context | Outcome |
|---|---|---|---|---|
| Tier 1 (Screening) | No chemical property or ecotoxicological data available | Quantitative Structure-Activity Relationship (QSAR) models (e.g., ECOSAR) [12] [11] | Priority setting, initial hazard identification, data gap filling for high-volume chemicals | Conservative hazard estimates to screen out low-risk substances |
| Tier 2 (Refined) | Some data available, requiring more accurate characterization | Read-Across from data-rich analogue substances [13] | Hazard assessment for chemicals with some experimental data on analogues | More reliable, substance-specific hazard characterization |
| Tier 3 (Complex) | Data-rich situations or specific research questions | Biologically-Based Models (e.g., Toxicokinetic-Toxicodynamic (TK-TD) models, Dynamic Energy Budget (DEB) models, Physiologically Based Models (PBMs)) [1] [11] | Derivation of specific reference points, extrapolation to population-level effects, understanding mechanisms | High-resolution risk estimates, often for specific ecosystems or populations |
This logical progression through tiers is outlined in the workflow below.
The Weight of Evidence (WoE) Framework: Integrating Multiple Lines of Evidence
A WoE approach is a structured process for transparently assembling, weighing, and integrating multiple, sometimes conflicting, strands of evidence to reach a conclusive assessment [10]. It is particularly critical when using in silico methods, as no single model is universally reliable.
The process involves systematically combining results from complementary in silico tools, such as statistical-based QSARs and expert rule-based systems, and potentially integrating them with existing in vitro or in vivo data [10] [3]. The convergence of predictions from multiple independent models increases confidence in the assessment, while conflicting results signal a need for more refined analysis or indicate high uncertainty.
Table 2: Key In Silico Tools for Integration in a Weight of Evidence Framework
| Tool Category | Specific Tool/Model | Function in WoE | Key Endpoints |
|---|---|---|---|
| QSAR Platforms | ECOSAR (Ecological Structure Activity Relationships) [12] | Predicts aquatic toxicity for screening; provides quantitative estimates for data-poor chemicals. | Acute and chronic toxicity to fish, daphnids, algae. |
| Expert Knowledge Systems | OECD QSAR Toolbox, Derek Nexus | Identifies structural alerts associated with toxicity; provides mechanistically-supported predictions. | Genotoxicity, skin sensitization, endocrine disruption. |
| Open-Access Databases | ECOTOX Knowledgebase [12], EPA CompTox Chemicals Dashboard [11] | Provides curated in vivo ecotoxicity data for validation of in silico predictions and read-across. | Experimental toxicity values for aquatic and terrestrial species. |
| Toxicokinetic Models | High-Throughput Toxicokinetic (HTTK) or generic one-compartment models [11] | Predicts internal doses from external exposures, linking in vitro bioactivity to in vivo relevance. | Bioavailability, metabolism, half-life. |
The following diagram illustrates the dynamic process of synthesizing evidence from these diverse tools.
Experimental Protocols for Key In Silico Methodologies
Protocol for Read-Across Assessment
Read-across is a powerful data-gap filling technique that requires a rigorous, transparent protocol to be acceptable for regulatory purposes [13].
- Define Target Substance: Clearly identify the substance for which data is missing (the "target").
- Identify Analogues: Systematically search for potential source analogues based on:
- Structural similarity (common functional groups, carbon chain length).
- Similarity in physical-chemical properties (e.g., log Kow, molecular weight).
- A common metabolic pathway or mode of action.
- Justify Analogue Selection: Provide a robust scientific rationale for the chosen analogues, demonstrating that the similarities are sufficient to support extrapolation for the specific endpoint in question.
- Data Collection and Evaluation: Gather all available experimental data for the source analogues for the target endpoint (e.g., ecotoxicity). Assess the reliability of these data.
- Address Uncertainties: Explicitly discuss any differences between the target and source analogues and how they might affect the prediction. This may involve providing a qualitative or quantitative uncertainty analysis.
- Documentation: Compile the entire assessment, including all data, justifications, and uncertainty analyses, in a complete and transparent report following guidelines from ECHA and OECD [13].
Protocol for a WoE Assessment Using QSARs
This protocol ensures in silico toxicological assessments are performed and evaluated in a consistent, reproducible, and well-documented manner [10].
- Endpoint and Model Selection: Precisely define the toxicological endpoint of interest (e.g., fish acute toxicity). Select at least two complementary QSAR methodologies—one statistical-based (e.g., from EPA's ECOSAR) and one expert rule-based (e.g., from OECD QSAR Toolbox) [10].
- Prediction and Documentation: Run the predictions for the target chemical. Document the exact software, version, and all input parameters used.
- Assess Applicability Domain (AD): For each model, evaluate whether the target chemical falls within its AD—the chemical space on which the model was built and for which it is reliable. If outside the AD, the prediction should be discounted or used with extreme caution.
- Evaluate Reliability and Relevance: Determine the reliability of the prediction (based on the model's validation and AD) and its relevance to the specific assessment context.
- Integrate Findings: Synthesize the results from the different models:
- If predictions are concordant and within the models' AD, confidence is high.
- If predictions are discordant, investigate the cause (e.g., different structural alerts, one model outside its AD). The assessment may require additional models or default to the more conservative prediction.
- Final Confidence Statement: Issue a final conclusion on the hazard, accompanied by a clear statement on the overall confidence level based on the relevance, reliability, and consistency of the integrated evidence [10].
Table 3: Key Research Reagent Solutions for In Silico ERA
| Tool Name | Type | Primary Function in Research |
|---|---|---|
| ECOSAR [12] | QSAR Software | Predicts acute and chronic toxicity of chemicals to aquatic organisms (fish, daphnids, green algae) based on chemical structure. |
| EPA CompTox Chemicals Dashboard [11] | Database | Provides access to curated physicochemical, toxicity, and exposure data for thousands of chemicals, supporting read-across and model development. |
| OECD QSAR Toolbox | Expert System | Identifies structural alerts, profiles metabolites, and facilitates grouping of chemicals for read-across to fill data gaps. |
| ECOTOX Knowledgebase [12] | Database | A curated repository of single-chemical ecological toxicity data for aquatic and terrestrial life, essential for validating in silico predictions. |
| OpenFoodTox [11] | Database | EFSA's open-source toxicological database on chemicals in food and feed, used for hazard identification and characterization. |
The future of in silico ERA lies in addressing increasingly complex challenges. Tiered and WoE approaches are fundamental to next-generation risk assessment (NGRA), which aims to holistically evaluate the impacts of multiple chemicals and multiple stressors (e.g., chemicals combined with temperature stress or pathogens) on living organisms and ecosystems [1] [11]. The development of landscape-based modeling approaches, which integrate geospatial data and landscape characteristics into exposure and effect assessments, represents the cutting edge of this field, moving risk assessment from a generic to a specific, systems-based context [1] [11]. The continued development and standardization of protocols for in silico methods, as surveyed in international initiatives [10], will be crucial for their wider regulatory acceptance and application. By firmly embedding tiered approaches and Weight of Evidence within problem formulation, the scientific community can ensure that in silico environmental risk assessment remains a robust, efficient, and transformative tool for protecting environmental health.
The Adverse Outcome Pathway (AOP) is a conceptual framework that systematically organizes existing knowledge concerning biologically plausible and empirically supported links between a molecular-level perturbation of a biological system and an adverse outcome at a level of biological organization relevant to regulatory decision-making [14]. The AOP framework provides a structure for capturing and representing the sequence of causal events that connect a molecular initiating event (MIE), such as the interaction of a chemical with a specific biological target, through a series of intermediate key events (KEs), to an adverse outcome (AO) of regulatory significance [15]. This framework facilitates greater integration and more meaningful use of mechanistic data in regulatory toxicology, potentially improving the efficiency and reliability of chemical safety assessment and environmental risk assessment [14].
Within the broader context of in silico environmental risk assessment research, AOPs play a pivotal role in supporting the transition from traditional chemical safety testing, which relies heavily on whole-animal studies, toward a more mechanistic and predictive approach [2]. By providing a structured representation of toxicity pathways, AOPs enable the development of computational models and in vitro testing strategies that can reduce reliance on animal testing, decrease costs, and accelerate the evaluation of chemical hazards [16] [2]. The framework supports a paradigm shift in toxicology toward greater utilization of mechanistic data for predicting adverse effects, which is particularly valuable for assessing the thousands of chemicals in commercial use with limited safety information [17].
Foundational Principles and Structure of AOPs
The development and application of AOPs are guided by five fundamental principles that ensure consistency and utility across the scientific community [14]:
AOPs are not chemical-specific: They describe generalizable biological pathways that can be initiated by any stressor (chemical, physical, or biological) capable of triggering the molecular initiating event.
AOPs are modular and composed of reusable components: The fundamental building blocks of AOPs are key events (KEs) and key event relationships (KERs), which can be shared across multiple AOPs.
An individual AOP is a pragmatic unit of development: A single linear sequence of KEs and KERs represents a manageable unit for initial development and evaluation.
Networks of AOPs are the functional unit of prediction: Most real-world scenarios involve interconnected AOPs that share common KEs and KERs, forming networks that better represent complex biological responses.
AOPs are living documents: They evolve over time as new scientific knowledge is generated, requiring periodic updates and refinements.
The core structural components of any AOP consist of [14] [16]:
- Molecular Initiating Event (MIE): The initial point of interaction between a stressor and a biological macromolecule (e.g., protein binding, receptor activation).
- Key Events (KEs): Measurable changes in biological state that are essential for progression along the pathway. These occur at different levels of biological organization (cellular, tissue, organ, organism).
- Key Event Relationships (KERs): Causal linkages between key events that describe how one event leads to another.
- Adverse Outcome (AO): An effect of regulatory significance at the individual or population level.
Visualizing the AOP Framework
The following diagram illustrates the generalized structure of an Adverse Outcome Pathway, showing the sequential progression from molecular initiation to adverse outcome:
Quantitative AOPs (qAOPs): From Qualitative to Quantitative Frameworks
While qualitative AOPs provide valuable conceptual frameworks, the development of quantitative AOPs (qAOPs) is essential for enabling predictive toxicology and risk assessment [16]. A qAOP incorporates mathematical models that quantitatively describe the relationships between key events, allowing for the prediction of the probability, timing, and severity of adverse outcomes based on the intensity or duration of exposure to a stressor [17]. The transition from qualitative AOPs to qAOPs represents a significant advancement in the field, as it enables more robust and reliable chemical safety assessments [16].
Methodological Approaches for qAOP Development
Several computational approaches have been employed to develop qAOPs, each with distinct advantages and applications [16]:
Response-Response Relationships: These involve fitting mathematical functions (e.g., regression models) to empirical data that describe the relationship between two adjacent key events. This approach is relatively straightforward but may lack biological mechanistic depth.
Biologically-Based Mathematical Modeling: This approach uses systems of ordinary differential equations to represent the underlying biological processes mechanistically. While more complex to develop, these models provide greater insight into the dynamics of the pathway.
Bayesian Networks (BNs): BNs are graphical models that represent probabilistic relationships among key events. They are particularly useful for handling uncertainty, integrating diverse data types, for modeling complex AOP networks with multiple branching pathways, as long as there are no feedback loops [16]. Dynamic Bayesian Networks can further incorporate temporal aspects.
Workflow for qAOP Development
The conversion of a qualitative AOP to a quantitative qAOP follows a systematic process [16] [17]:
Comprehensive Literature Review: A thorough examination of existing scientific literature to gather qualitative and quantitative data relevant to the AOP components.
Data Extraction and Categorization: Quantitative data suitable for model development is extracted and categorized. Ideally, this includes studies that measure multiple key events simultaneously.
Model Structure Definition: Based on the AOP structure and available data, an appropriate mathematical modeling approach is selected.
Parameter Estimation: Model parameters are estimated using available experimental data through statistical fitting procedures.
Model Evaluation and Validation: The qAOP model is tested against independent datasets not used in model development to assess its predictive performance.
Uncertainty Analysis: Sources of uncertainty in the model predictions are identified and quantified.
Implementation and Application: The validated qAOP is implemented in user-friendly tools and applied for chemical risk assessment.
Quantitative Modeling Techniques Used in qAOP Development
Table 1: Summary of Quantitative Modeling Approaches for qAOPs
| Modeling Approach | Key Features | Data Requirements | Advantages | Limitations |
|---|---|---|---|---|
| Response-Response Relationships [16] | Statistical fitting of functions between adjacent KEs | Paired measurements of adjacent KEs | Simple to implement; Minimal computational resources | Limited extrapolation capability; Less biological mechanistic depth |
| Biologically-Based Mathematical Models [16] | Systems of differential equations representing biological mechanisms | Time-course data; Kinetic parameters | Mechanistic insight; Good extrapolation potential | High data requirements; Computational complexity |
| Bayesian Networks (BNs) [16] [15] | Probabilistic graphs representing relationships between KEs | Conditional probability distributions | Handles uncertainty; Integrates diverse data types | Cannot model feedback loops without extensions |
AOP Case Studies in Environmental Risk Assessment
Case Study 1: Acetylcholinesterase Inhibition Leading to Neurodegeneration
AOP 281, "Acetylcholinesterase Inhibition Leading to Neurodegeneration," provides a well-characterized example of AOP development and the challenges associated with quantitative implementation [16]. This AOP describes the sequence of events through which inhibition of acetylcholinesterase (AChE) can ultimately lead to neurodegenerative effects:
The molecular initiating event is AChE inhibition, resulting in an excess of acetylcholine (ACh) in the synapse. This build-up of ACh leads to overactivation of muscarinic acetylcholine receptors (mAChR) within the brain, initiating local (focal) seizures. Through subsequent glutamate release and activation of NMDA receptors, the excitotoxicity propagates, leading to elevated intracellular calcium levels, status epilepticus, and ultimately cell death and neurodegeneration [16].
The qAOP development for this pathway faced several challenges, including the availability of quantitative data amenable to model development, the lack of studies measuring multiple key events simultaneously, and issues regarding model accessibility and transferability across platforms [16]. The case study highlighted the importance of improving key event and key event relationship descriptions in the AOP Wiki to facilitate the transition from qualitative to quantitative AOPs.
Case Study 2: Aromatase Inhibition Leading to Reproductive Dysfunction
AOP 25, "Aromatase Inhibition Leading to Reproductive Dysfunction," represents one of the more advanced AOPs with a developed quantitative component [16]. This AOP describes how inhibition of the aromatase enzyme, which converts androgens to estrogens, can lead to impaired reproduction in fish. The development of a qAOP for this pathway demonstrated that even AOPs with primarily textual descriptions in their quantitative understanding sections can be successfully converted to quantitative models [16]. This case study illustrates the potential for qAOPs to support predictive risk assessment for endocrine-disrupting chemicals in aquatic environments.
Visualizing the Acetylcholinesterase Inhibition AOP
The following diagram details the specific key events and relationships in AOP 281 (Acetylcholinesterase Inhibition Leading to Neurodegeneration), including the positive feedback loop that exacerbates the adverse outcome:
AOPs in Regulatory Science and Chemical Safety Assessment
The AOP framework plays an increasingly important role in modernizing chemical safety assessment and environmental risk assessment paradigms. By providing a structured representation of toxicity pathways, AOPs support several key applications in regulatory science [14] [15] [2]:
Integrated Approaches to Testing and Assessment (IATA): AOPs provide the scientific basis for developing integrated testing strategies that efficiently combine non-animal methods, such as high-throughput in vitro assays and in silico models, for chemical hazard characterization.
Chemical Prioritization and Screening: AOPs enable the development of targeted testing strategies for identifying chemicals with specific modes of action, facilitating more efficient prioritization of chemicals for further testing.
Extrapolation Across Species: By focusing on conserved biological pathways, AOPs support extrapolation of effects across species, which is particularly valuable for ecological risk assessment.
Quantitative Risk Assessment: qAOPs provide a foundation for developing predictive models that can estimate the probability and magnitude of adverse effects at environmentally relevant exposure concentrations.
The use of AOPs in regulatory decision-making is facilitated by international collaborations, particularly through the Organisation for Economic Co-operation and Development (OECD) AOP Development Programme, which oversees the formal review and endorsement of AOPs [16]. This harmonized approach ensures that AOPs developed by the scientific community meet agreed-upon standards of scientific rigor and reliability for application in regulatory contexts.
Experimental Protocols and Methodologies for AOP Development
Weight of Evidence Assessment for AOP Development
The establishment of scientifically credible AOPs requires a systematic weight of evidence (WoE) assessment based on modified Bradford-Hill criteria [16]. This assessment evaluates three fundamental aspects of each Key Event Relationship (KER):
Biological Plausibility: Evaluation of the strength of evidence supporting a causal relationship between key events based on current understanding of biological system.
Empirical Support: Assessment of the extent and consistency of experimental observations demonstrating that a change in the upstream key event leads to an appropriate change in the downstream key event.
Quantitative Understanding: Evaluation of the extent to which dose-response, temporal, and incidence relationships between key events are understood.
The WoE assessment is typically documented in the AOP Wiki (https://aopwiki.org/), the central repository for AOP knowledge, which provides standardized forms for capturing and evaluating the evidence supporting each KER [16].
Essential Research Reagents and Tools for AOP-Related Research
Table 2: Key Research Reagents and Tools for AOP Development and Validation
| Reagent/Tool Category | Specific Examples | Research Application in AOP Context |
|---|---|---|
| In Vitro Bioassays [2] | Acetylcholinesterase activity assays; Aromatase inhibition assays; Receptor binding assays | Measuring Molecular Initiating Events (MIEs) and cellular-level Key Events (KEs) for high-throughput chemical screening |
| Biomarker Assays [15] | Oxidative stress markers; Hormone level measurements; Specific protein biomarkers (e.g., Vitellogenin) | Quantifying changes in intermediate Key Events (KEs) to track progression along the AOP |
| 'Omics Technologies [15] | Transcriptomics; Proteomics; Metabolomics | Discovering novel Key Events (KEs) and providing system-wide evidence for biological plausibility of Key Event Relationships (KERs) |
| Computational Models [16] [2] | Physiologically Based Kinetic (PBK) models; Bayesian Networks; Quantitative Structure-Activity Relationship (QSAR) models | Developing Quantitative AOPs (qAOPs); Extrapolating from in vitro to in vivo exposures and across species |
| Reference Chemicals [16] | Specific agonists/antagonists for molecular targets; Chemicals with well-characterized modes of action | Establishing biological plausibility and empirical support for Key Event Relationships (KERs) during AOP development |
Challenges and Future Directions in AOP Development
Despite significant progress in AOP development and application, several challenges remain to be addressed [16] [2] [17]:
Quantitative Data Gaps: Many existing AOPs lack the quantitative data necessary for developing robust qAOP models. There is a particular need for studies that measure multiple key events simultaneously across different biological levels.
AOP Network Complexity: Most adverse outcomes result from multiple interconnected pathways rather than simple linear sequences. Modeling these complex networks presents both conceptual and computational challenges.
Biological Variability: Accounting for inter-individual and cross-species variability in quantitative AOP models remains difficult but is essential for accurate risk assessment.
Technical Barrier: Model accessibility and transferability across different platforms and research groups need improvement to facilitate wider adoption of qAOPs.
Dynamic and Feedback Processes: Many biological systems include feedback mechanisms and adaptive responses that are not yet fully captured in current AOP frameworks.
Future directions in AOP research include the development of more sophisticated computational approaches for qAOP modeling, enhanced international collaboration for populating the AOP knowledgebase, and stronger integration of AOPs into regulatory decision-making processes [16] [2] [17]. The continued evolution of the AOP framework is expected to play a crucial role in advancing the goals of 21st-century toxicology and in silico environmental risk assessment, ultimately leading to more efficient, cost-effective, and human-relevant chemical safety assessment.
For decades, animal testing has served as the cornerstone of regulatory safety assessment for pharmaceuticals, pesticides, and industrial chemicals. However, a growing body of evidence demonstrates significant scientific and economic limitations in this approach. Animal models are poor predictors of human toxicity, with analysis suggesting they are "little better than what would result merely by chance—or tossing a coin—in providing a basis to decide whether a compound should proceed to testing in humans" [18]. Approximately 89% of novel drugs fail human clinical trials, with about half of these failures due to unanticipated human toxicity not detected in animal studies [18].
The economic implications are equally staggering. Rodent testing in cancer therapeutics adds an estimated 4 to 5 years to drug development and costs $2 to $4 million per compound. For industrial toxicity testing, completing all required animal studies for a single pesticide takes approximately 10 years and $3 million [18]. Compared with in vitro testing, animal tests range from 1.5× to >30× more expensive [18]. These scientific and economic limitations have catalyzed the development and adoption of New Approach Methodologies (NAMs), particularly in silico methods, which offer faster, cost-effective, and increasingly accurate alternatives for environmental risk assessment [19].
Limitations of Animal Testing: Scientific and Economic Concerns
Scientific Concordance and Predictive Value
The scientific foundation of animal testing is undermined by poor concordance with human outcomes. A review of 221 animal experiments found agreement in human studies just 50% of the time—essentially random chance [18]. Analysis of the U.S. National Toxicology Program concluded that toxicities other than carcinogenesis were not reproducible between rats and mice, between sexes, or compared with historic control animals [18].
Table 1: Concordance Between Animal Studies and Human Outcomes
| Study Focus | Number of Studies/Chemicals Analyzed | Concordance Rate | Key Finding |
|---|---|---|---|
| Animal experiment reproducibility | 221 experiments | 50% | Agreement with human studies no better than chance |
| Mouse to rat toxicity prediction | 37 chemicals | 55.3% (long-term), 44.8% (short-term) | Little better than random prediction |
| Pharmaceutical failure rate | N/A | 89% overall failure rate | ~50% due to unanticipated human toxicity |
| Post-marketing safety issues | 93 serious adverse outcomes | 19% | Only 19% identified in preclinical animal studies |
Notable Cases of Failed Prediction
Historical evidence reveals numerous concerning failures of animal studies to predict human toxicity:
- Vioxx (Merck): Withdrawn after release despite animal testing, associated with an estimated 88,000 heart attacks and 38,000 deaths [18]
- Thalidomide: Caused devastating phocomelia in 20,000-30,000 infants despite testing in 10 strains of rats, 11 breeds of rabbits, 2 breeds of dogs, 3 strains of hamsters, 8 species of primates, and various other species [18]
- TGN1412: An antibody given at 1/500th the dose found safe in animal studies rendered all 6 human volunteers critically ill within minutes [18]
- Fialuridine: Caused the deaths of 5 volunteers during phase II clinical trials despite being safe in multiple animal species at hundreds of times higher doses [18]
The Economic Burden of Animal Testing
The economic impact of reliance on animal testing extends beyond direct costs to include opportunity costs from abandoned potentially beneficial compounds and delayed market access:
- Direct costs: Pesticide testing can cost up to $9,919,000 overall, with chronic toxicity studies taking up to 2 years [2]
- Opportunity costs: Drugs falsely identified as toxic in animal studies are often abandoned, though the magnitude of this error is unknown as these compounds rarely proceed to human testing [18]
- Delay costs: Compassionate human use of ganciclovir demonstrated efficacy and safety in more patients than required for phase I/II trials, but FDA delayed approval for 4 years due to lack of animal studies [18]
In Silico Methodologies: A New Paradigm for Risk Assessment
Defining In Silico Environmental Risk Assessment
In silico environmental risk assessment (ERA) represents a paradigm shift from traditional animal-based testing to computational approaches that predict chemical behavior, toxicity, and environmental fate. These methodologies use computer simulations, mathematical models, and computational chemistry to evaluate potential hazards [20]. The European Commission encourages the use of validated in silico techniques such as (Q)SAR models as part of reduction, refinement, and replacement strategies for animal use [4].
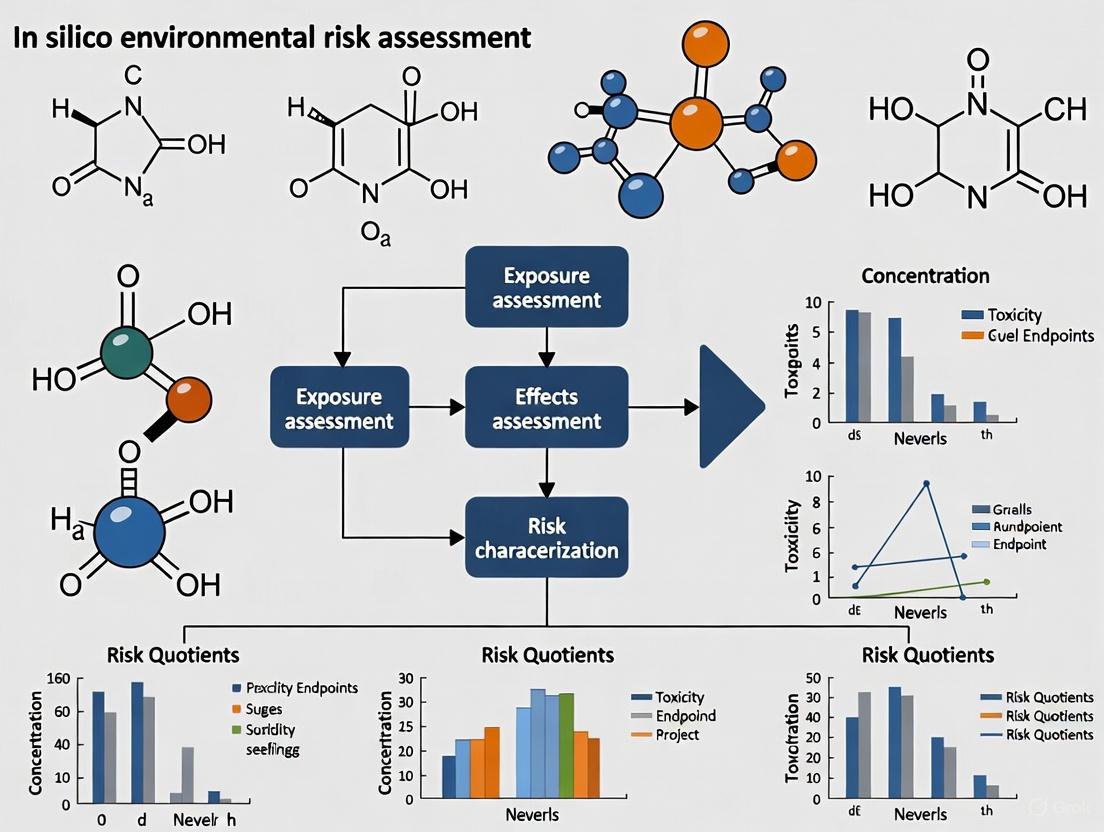
The framework for ERA typically involves four distinct steps: hazard identification, exposure assessment, toxicity assessment, and risk characterization [2]. In silico tools can contribute to each of these steps, creating an integrated approach that minimizes animal use while providing robust safety data.
Key In Silico Approaches and Tools
Quantitative Structure-Activity Relationship (QSAR) Models
QSAR models correlate chemical structure with biological activity or properties using mathematical relationships. These models operate on the principle that structurally similar chemicals have similar properties or activities. The OECD QSAR Toolbox is a freely available software application that supports reproducible chemical hazard assessment, offering functionalities for retrieving experimental data, simulating metabolism, and profiling properties of chemicals [21].
Read-Across and Category Approaches
Read-across is a data gap filling technique where properties of a data-rich "source" chemical are used to predict the same properties for a similar, data-poor "target" chemical. The OECD Toolbox facilitates this approach by helping to find structurally and mechanistically defined analogues and chemical categories [21]. Tools like RAXpy enable read-across based on structural, biological, and metabolic similarities [22].
Exposure Assessment Models
For pesticide exposure assessment, models like AGDISP (AGricultural DISPersal model) effectively monitor pesticide deposition and spray drift, successfully tracking atrazine drift up to 400 meters from application sites [2]. These tools help characterize environmental fate and potential human exposure without extensive field studies.
Integrated Testing Strategies
The most powerful applications combine multiple in silico approaches through Integrated Approaches for Testing and Assessment (IATA), which combine different data sources to conclude on chemical toxicity [19]. IATA frameworks integrate and weigh all relevant existing evidence while guiding targeted new data generation.
Table 2: Key In Silico Tools for Environmental Risk Assessment
| Tool Name | Primary Function | Access | Key Features |
|---|---|---|---|
| OECD QSAR Toolbox [21] | Chemical hazard assessment | Free | 63 databases with 155K+ chemicals and 3.3M+ experimental data points; read-across and category formation |
| VERMEER Cosmolife [22] | Cosmetic risk assessment | Not specified | Evaluation of cosmetic ingredients and detailed investigation of risk scenarios |
| SILIFOOD [22] | Food contact material assessment | Free | Fast risk assessment of non-evaluated Food Contact Material substances |
| AGDISP [2] | Pesticide spray drift prediction | Not specified | Predicts pesticide deposition and drift in agricultural applications |
| BeeTox [2] | Bee toxicity prediction | Not specified | Graph attention convolutional neural network distinguishing bee-toxic chemicals with 83.7% accuracy |
| ToxEraser FCM [22] | Food contact material identification | Free | Identifies risky food contact materials and suggests safer alternatives |
Quantitative Benefits: Cost and Time Savings of In Silico Methods
The transition to in silico methods offers substantial quantitative benefits in both cost reduction and testing efficiency. Analysis of 261 compounds demonstrated that in silico methods could eliminate the use of 0.1–0.15 million test animals and save $50–70 billion [2]. This represents a paradigm shift in the economics of toxicity testing.
A framework for cost-effectiveness analyses of toxicity tests that accounts for cost, duration, and uncertainty uses the metric of "cost per correct regulatory decision" [23]. This approach recognizes that either a fivefold reduction in cost or duration can be a larger driver of optimal methodology selection than a fivefold reduction in uncertainty, particularly for simpler regulatory decisions [23].
Table 3: Economic Comparison: Animal Testing vs. In Silico Approaches
| Parameter | Animal Testing | In Silico Approaches | Advantage Ratio |
|---|---|---|---|
| Pesticide registration | ~10 years [18] | Significantly reduced (case-dependent) | >5× faster [23] |
| Pesticide testing cost | Up to $9,919,000 [2] | Dramatically reduced | 1.5× to >30× cost savings [18] |
| Cancer therapeutic rodent testing | $2-4 million [18] | Cost of software and computational resources | Potentially orders of magnitude |
| Animal use per 261 compounds | 100,000-150,000 animals [2] | Minimal to none | Near total replacement |
| Drug development timeline impact | Adds 4-5 years [18] | Can be integrated early and rapidly | Significant acceleration |
Implementation Framework: Protocols for In Silico ERA
Systematic Workflow for In Silico Assessment
A robust in silico environmental risk assessment follows a structured workflow that ensures scientific rigor and regulatory acceptance. The CADASTER project (Case studies on the Development and Application of in-Silico Techniques for Environmental hazard and Risk assessment) exemplifies this approach through its focus on collecting existing data and models, assessing quality of toxicity data, developing new QSAR models, and characterizing uncertainty [4].
Experimental Protocols for Model Development and Validation
Data Collection and Curation (Task 2.1 from CADASTER)
The foundation of any in silico assessment is robust data collection. The protocol includes:
- Literature search for endpoints relevant to environmental risk assessment
- Database mining of existing databases on risk and hazard assessment parameters
- Data quality assessment to characterize variability and uncertainty in available data
- Supplementation with new experimental data where essential for model development [4]
Model Development and Validation
The development of new QSAR models follows rigorous protocols:
- Descriptor calculation: Breaking chemical structures into fragments or calculating molecular descriptors
- Dataset splitting: Dividing data into training, calibration, and test sets to avoid overtraining [22]
- Model training: Using algorithms such as counter-propagation artificial neural networks (CPANN) or self-organising maps (SOM) [22]
- Applicability domain assessment: Defining the chemical space where the model provides reliable predictions [4]
- Validation: Assessing predictive power using external test sets and statistical measures
Integrated Risk Assessment Protocol
For comprehensive environmental risk assessment of pharmaceuticals and transformation products, researchers have successfully implemented protocols that:
- Evaluate biodegradability using predictive software
- Assess carcinogenicity and mutagenicity through computational models
- Apply chemometric analyses (HCA and PCA) to enhance result interpretation [24]
- Prioritize compounds of concern based on multiple endpoints
Successful implementation of in silico approaches requires access to specialized software, databases, and computational resources. This toolkit enables researchers to replace animal testing while maintaining scientific rigor.
Table 4: Essential Research Reagent Solutions for In Silico Risk Assessment
| Tool Category | Specific Tools | Key Function | Regulatory Relevance |
|---|---|---|---|
| QSAR Platforms | OECD QSAR Toolbox [21], SARpy, CORAL, QSARpy [22] | Develop and apply QSAR models for various endpoints | Accepted in REACH assessments [4] |
| Read-Across Tools | RAXpy [22], Read-Across functionality in OECD Toolbox [21] | Identify analogues and fill data gaps using similar compounds | Recommended by EFSA and ECHA for genotoxicity assessment [19] |
| Exposure Models | AGDISP [2], TOXSWA | Predict environmental fate and concentration of chemicals | Used in pesticide registration and environmental monitoring |
| Toxicity Prediction | BeeTox [2], aiQSAR [22] | Predict specific toxicity endpoints for environmental organisms | Supports pesticide risk assessment and prioritization |
| Metabolic Simulators | Metabolism simulators in OECD Toolbox [21] | Predict metabolic pathways and transformation products | Critical for assessing persistence and bioaccumulation |
| Reporting Tools | QMRF reporting, Data matrix wizard in OECD Toolbox [21] | Generate transparent assessment reports | Essential for regulatory acceptance and review |
Validation and Regulatory Acceptance: Building Confidence in In Silico Methods
The regulatory acceptance of in silico methods depends on rigorous validation and demonstration of reliability. The CADASTER project addressed this through several key activities: development of methodologies for assessing applicability domains of models, characterization of uncertainty and variability, and sensitivity analysis of individual models [4].
Current regulatory frameworks increasingly recognize the value of in silico approaches. The REACH system requires that non-animal methods should be used for the majority of tests in the 1-10 tonne band [4]. Similarly, the European animal testing bans for cosmetics (Regulation No 1223/2009) have accelerated development and validation of alternative approaches [25].
For complex endpoints, regulatory agencies including the U.S. EPA, EFSA, and ECHA have been developing frameworks to implement and use NAMs for regulatory applications [19]. The integration of in silico predictions with other data sources through "weight of evidence" approaches allows for confident decision-making even for complex endpoints.
The field of in silico environmental risk assessment continues to evolve rapidly, with several promising developments on the horizon. The integration of artificial intelligence and machine learning approaches is enhancing predictive capabilities, while initiatives for data FAIRification (Findable, Accessible, Interoperable, and Reusable) are improving data quality and accessibility [19].
Future advancements need to address several critical challenges, including better consideration of environmental exposure concentrations, interactions among mixtures of contaminants, and development of in silico models specifically tailored for ERA of emerging contaminants like pharmaceuticals [20]. Additionally, increasing the regulatory acceptance and standardization of these methods will be crucial for wider adoption.
In conclusion, in silico methods represent a scientifically robust and economically viable alternative to traditional animal testing for environmental risk assessment. The compelling evidence of cost savings, coupled with improved efficiency and growing regulatory acceptance, positions these approaches as key drivers in overcoming the limitations of animal testing. As computational power increases and algorithms become more sophisticated, the role of in silico methods is poised to expand further, ultimately leading to more predictive, cost-effective, and ethical chemical safety assessment.
Global chemical regulatory frameworks are actively driving a pivotal transformation in environmental and human health risk assessment. Spearheaded by REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) in the European Union, in conjunction with international standards from the Organisation for Economic Co-operation and Development (OECD) and specific sectoral regulations like those for cosmetics, regulators are increasingly mandating a move away from traditional animal-based testing. This shift is underpinned by the parallel goals of enhancing ethical standards and embracing more scientifically advanced, human-relevant methodologies. The core of this transformation lies in the adoption of New Approach Methodologies (NAMs), which include in silico (computational) tools, in vitro assays, and advanced data integration techniques. These approaches are recognized not merely as alternatives but as superior pathways for generating robust, efficient, and mechanistically insightful safety data, thereby firmly establishing the regulatory context that encourages and validates in silico environmental risk assessment research [26] [27] [28].
Regulatory Frameworks as Catalysts for Change
REACH and the Push for NAMs
The REACH regulation establishes a comprehensive framework for chemicals in the EU, creating a direct impetus for the development and use of alternative assessment methods. Its core processes—registration, evaluation, and restriction—increasingly incorporate provisions for non-animal data.
- Registration and Data Gaps: REACH requires that chemical substances produced or imported in quantities exceeding one tonne per year be registered with the European Chemicals Agency (ECHA). For this process, companies must identify and manage the risks linked to the substances [29]. ECHA's 2025 report on "Key Areas of Regulatory Challenge" explicitly outlines research needs where NAMs are critical, including for assessing neurotoxicity, immunotoxicity, endocrine disruption, and environmental endpoints like persistence and bioaccumulation [27].
- Analogical Reasoning and Read-Across: REACH explicitly endorses the use of read-across, where data from a tested "source" substance is used to predict the properties of a similar "target" substance. ECHA notes that NAMs can strengthen this process by "generating toxicokinetic and toxicodynamic data for candidate analog substances," thereby validating the assumptions of similarity and filling data gaps without additional animal testing [27].
- Specific Endpoint Challenges: The regulatory body highlights specific areas where in silico and in vitro methods are poised to reduce vertebrate testing. For instance, for fish toxicity, both short-term and long-term, NAMs are under development to predict adverse outcomes without the use of live animals. Similarly, for carcinogenicity, which traditionally relies on two-year rodent bioassays, there is an active push to "use alternative and new methods to speed up the detection of carcinogens" [27].
Cosmetic Regulations: A Vanguard for Non-Animal Approaches
Cosmetic regulations have been the most forceful drivers of the transition to NAMs, effectively creating a regulatory environment where non-animal methods are a necessity, not an option.
- The EU Animal Testing Ban: EU Cosmetic Regulation (EC) No 1223/2009 explicitly prohibits animal testing for finished cosmetic products and their ingredients [30] [31]. This ban has made the development and application of NAMs a commercial and regulatory imperative for the entire cosmetics industry operating in the EU market.
- Next-Generation Risk Assessment (NGRA): The cosmetics industry, in response to these regulations, is pioneering NGRA. This is defined as a "human-relevant, exposure-led, hypothesis-driven risk assessment approach, designed to prevent harm" [26] [28]. NGRA relies heavily on a suite of NAMs—including in silico and in vitro tools—to perform safety assessments without animal data. The International Cooperation on Cosmetics Regulation (ICCR) is actively working to define best practices to advance the regulatory acceptance of these methodologies [26].
- Global Harmonization of Cosmetic Rules: The influence of the EU's approach is global. The United States' Modernization of Cosmetics Regulation Act of 2022 (MoCRA) mandates safety substantiation for cosmetic products, a requirement that inherently promotes the use of advanced, efficient assessment techniques [30] [32]. Similarly, Brazil recently passed a law prohibiting the use of animals in cosmetic testing, further expanding the global market for which non-animal safety data is required [30].
OECD and the International Validation of Methods
The OECD plays a critical role in the international harmonization of chemical safety testing guidelines. The adoption of an OECD Test Guideline (TG) is a key step for a method to achieve widespread regulatory acceptance. The ongoing development and updating of TGs for NAMs provide the essential, globally recognized protocols that lend credibility and reliability to data generated through in silico and in vitro means, thereby facilitating their use in regulatory dossiers submitted under frameworks like REACH [33] [27].
Table 1: Regulatory Drivers for In Silico and NAM Adoption Across Frameworks
| Regulatory Framework | Key Mechanism | Impact on In Silico/NAM Adoption |
|---|---|---|
| REACH (EU) | Requirement for data on thousands of chemicals; endorsement of read-across [27]. | Creates a necessity to fill data gaps efficiently. Promotes use of (Q)SAR and other computational tools to meet information requirements. |
| EU Cosmetics Regulation | Full ban on animal testing for cosmetics [30] [31]. | Makes NAMs the only viable pathway for safety assessment, driving innovation in NGRA. |
| MoCRA (USA) | Requirement for safety substantiation of cosmetic products [30] [32]. | Encourages industry to adopt modern, efficient safety assessment methodologies, including in silico approaches. |
| OECD Guidelines | International validation and standardization of test methods [33]. | Provides the essential regulatory legitimacy and interoperability for non-animal methods across jurisdictions. |
Foundational In Silico Methodologies and Protocols
The regulatory push has accelerated the refinement and application of specific computational methodologies. These tools are integral to modern chemical safety and risk assessment pipelines.
Predictive Models for Transformation Products (TPs)
Understanding a chemical's environmental fate, particularly the formation and toxicity of TPs, is a critical aspect of a holistic risk assessment. In silico methods offer efficient solutions for prioritization and screening [33].
Rule-Based Models: These models are grounded in expert-curated, mechanistic reaction rules derived from experimental studies. They predict transformation pathways (e.g., hydroxylation, oxidation) by applying these predefined rules to a parent compound's structure. Their key strength is high interpretability, but they are limited to predicting known transformations [33].
- Protocol Example: Using a tool like
enviPathto predict biotic degradation pathways.- Input: Prepare the SMILES (Simplified Molecular-Input Line-Entry System) string of the parent compound.
- Parameter Setting: Select the desired transformation library (e.g., "biotic" for microbial degradation).
- Execution: Run the prediction algorithm to generate a tree of potential transformation products.
- Output Analysis: Review the predicted TPs and the proposed transformation pathways. The results are typically presented as a combination of chemical structures and reaction descriptions.
- Protocol Example: Using a tool like
Machine Learning (ML) Models: These data-driven models can uncover complex, non-linear relationships between chemical structure and transformation potential or toxicity. They are trained on large datasets of chemical properties and biological activities. While powerful and flexible, their "black-box" nature can sometimes hinder mechanistic interpretation [33].
- Protocol Example: Training an ML model to predict acute aquatic toxicity.
- Data Curation: Compile a high-quality dataset of chemical structures and associated experimental toxicity values (e.g., LC50 for fish).
- Descriptor Calculation: Generate numerical molecular descriptors (e.g., topological, electronic, physicochemical) for all compounds in the dataset.
- Model Training & Validation: Use an algorithm (e.g., Random Forest, Support Vector Machine) to learn the relationship between descriptors and toxicity. The dataset is typically split into training and validation sets to assess model performance and prevent overfitting.
- Prediction: Apply the trained model to new chemicals to estimate their toxicity.
- Protocol Example: Training an ML model to predict acute aquatic toxicity.
Integrated Workflows: The most advanced approaches combine rule-based and ML methods. Quantitative Structure-Activity Relationship (QSAR) models serve as a bridge, as they can be built using expert-defined descriptors or trained via ML. Similarly, read-across is increasingly enhanced by ML to identify optimal analogue substances and improve predictive accuracy [33].
The following diagram illustrates a typical computational workflow for the in silico assessment of transformation products, integrating both prediction and hazard evaluation:
Diagram: A computational workflow for predicting and prioritizing transformation products (TPs) for environmental risk assessment.
The reliability of any in silico model is contingent on the quality and breadth of its underlying data. Key resources for TP and toxicity data include:
- NORMAN Suspect List Exchange (NORMAN-SLE): A collaborative repository of suspect lists for screening chemical substances in the environment [33].
- PubChem: A public database of chemical molecules and their activities, which includes a "Transformations" section with parent-TP mappings [33].
- enviPath: A resource for predicting microbial biodegradation pathways and storing experimental data [33].
It is critical to note that while large language models (LLMs) might be prompted to propose TPs, they are not based on curated chemical rules and "should be treated with caution," as they may generate plausible but false information. Expert-curated databases are the recommended source [33].
The Scientist's Toolkit: Key Reagents and Research Solutions
Implementing a modern, in silico-driven risk assessment strategy requires a suite of computational and experimental tools. The table below details key components of this toolkit.
Table 2: Essential Research Tools for Next-Generation Risk Assessment
| Tool Category | Example / Solution | Function in Risk Assessment |
|---|---|---|
| Computational Prediction Platforms | enviPath, BioTransformer, OECD QSAR Toolbox |
Predicts environmental transformation pathways and metabolites of parent compounds [33]. |
| Toxicity Prediction Models | (Q)SAR models, molecular docking simulations, AOP (Adverse Outcome Pathway) networks | Forecasts key toxicological endpoints (e.g., mutagenicity, endocrine activity) from chemical structure [33] [28]. |
| Data Curation & Analysis | NORMAN-SLE, PubChem Transformations, ShinyTPs | Provides curated data on known transformation products and supports text-mining for literature-derived TP information [33]. |
| Exposure Assessment | ECHA Use Maps, SPERCs (Specific Environmental Release Categories), SCEDs (Specific Consumer Exposure Determinants) | Provides standardized exposure scenarios for workers, consumers, and the environment for use in Chemical Safety Assessments (CSAs) under REACH [34]. |
| Integrated Workflow Software | Chesar (Chemical Safety Assessment and Reporting tool) |
Enables companies to conduct, manage, and report their chemical safety assessments in a standardized and efficient manner [34]. |
| In Vitro Assays (for NGRA) | Transcriptomics, high-throughput screening assays, in vitro toxicokinetics | Generates human-relevant biological effect data to be integrated with in silico predictions and exposure data in a weight-of-evidence approach [26] [28]. |
The regulatory landscape, shaped by REACH, OECD, and groundbreaking cosmetic regulations, has unequivocally moved from merely accepting in silico methods to actively encouraging their development and application. The future of environmental risk assessment lies in integrated testing strategies that seamlessly combine in silico predictions, in vitro data, and human exposure information within a Next-Generation Risk Assessment (NGRA) framework. This approach is not only more ethical but also more scientifically relevant, efficient, and protective of human health and the environment. For researchers and drug development professionals, proficiency in these methodologies is no longer a niche specialty but a core competency required to navigate global regulatory requirements and contribute to the development of safer, more sustainable chemicals and products.
Tools of the Trade: Key In Silico Models and Their Real-World Applications
Quantitative Structure-Activity Relationship (QSAR) modeling represents a cornerstone of computational toxicology and environmental risk assessment. These in silico models predict the biological activity or toxicity of chemicals based on their molecular structure, utilizing statistical and machine learning methods to establish relationships between chemical descriptors and biological endpoints. As regulatory agencies increasingly advocate for New Approach Methodologies (NAMs) to reduce animal testing, QSAR models have gained significant importance for supporting safety assessment of consumer products, pharmaceuticals, and environmental contaminants. This technical guide examines the fundamental principles, development methodologies, validation frameworks, and applications of QSAR modeling, with particular emphasis on environmental risk assessment contexts. The document also explores emerging trends, including the integration of artificial intelligence and knowledge-based approaches that enhance predictive capabilities beyond traditional structure-based paradigms.
In silico environmental risk assessment (ERA) represents a paradigm shift in how scientists evaluate the potential hazards of chemicals in the environment. As a fundamental component of this approach, QSAR modeling enables researchers to predict the environmental fate and toxicological effects of chemicals without exhaustive laboratory testing. The foundation of QSAR rests on the principle that chemical structure determines biological activity—a concept formally established by Corwin Hansch in 1962 but with roots extending back to earlier work on linear free-energy relationships by Hammett and others [35].
The regulatory landscape has increasingly embraced QSAR methodologies. The U.S. Food and Drug Administration's 2025 Roadmap to Reducing Animal Testing in Preclinical Safety Studies emphasizes the adoption of NAMs, while the FDA Modernization Act 3.0 further supports this transition by modernizing toxicological assessment requirements [36]. Similarly, the European Union's REACH regulation promotes the use of QSAR to fill data gaps, particularly following bans on animal testing for cosmetics [37]. These developments position QSAR as an essential tool for addressing the thousands of chemicals requiring assessment while reducing reliance on animal studies and containing costs.
Environmental risk assessment for chemicals like pesticides exemplifies the value of QSAR approaches. Traditional testing can cost nearly $10 million and take up to two years for a single compound, whereas in silico methods offer rapid, cost-effective alternatives that can potentially save 50-70 billion dollars and reduce animal use by 100,000-150,000 for assessing 261 compounds [2]. For pesticide risk assessment, QSAR models help characterize environmental behavior, exposure potential, and ecological effects across aquatic, terrestrial, and soil compartments.
Fundamental Principles of QSAR Modeling
Historical Development and Theoretical Foundation
QSAR modeling has evolved significantly since its inception more than fifty years ago. The field originated from physical organic chemistry, particularly the work of Louis Hammett who established linear free-energy relationships to explain substituent effects on chemical reactivity. Hansch and Fujita's pioneering research in the early 1960s formalized QSAR by demonstrating that biological activity could be correlated with physicochemical parameters through mathematical equations [35]. Their approach incorporated hydrophobicity (measured by octanol-water partition coefficients, log P) alongside electronic and steric parameters, establishing the Hansch equation that remains influential today.
The fundamental QSAR paradigm operates on the similarity principle—the concept that structurally similar compounds tend to have similar biological properties [38]. This principle enables the prediction of activities for untested compounds based on their structural resemblance to chemicals with known activity profiles. However, this principle has limitations, particularly when minor structural modifications result in significant toxicity changes, as exemplified by the drug pair ibuprofen (generally safe) and ibufenac (withdrawn due to hepatotoxicity), which differ by only a single methyl group [36].
Key Components of QSAR Models
All QSAR models comprise three essential components: (1) molecular descriptors that numerically encode structural and physicochemical properties; (2) an algorithm that establishes the relationship between descriptors and biological activity; and (3) a defined applicability domain that specifies the model's scope and limitations.
Molecular descriptors quantify aspects of molecular structure and properties, including:
- Physicochemical descriptors: Hydrophobicity (log P), electronic properties (sigma constants), steric parameters
- Topological descriptors: Molecular connectivity indices, fingerprint patterns
- Geometric descriptors: Molecular shape, size, surface areas
- Quantum chemical descriptors: HOMO/LUMO energies, molecular orbital properties
Algorithm selection depends on the modeling context, with options ranging from traditional regression methods to advanced machine learning approaches:
Table 1: Machine Learning Algorithms for QSAR Development
| Algorithm | Complexity | Applicability | Interpretability |
|---|---|---|---|
| k-Nearest Neighbors (KNN) | Low | Small datasets, similarity-based | High |
| Logistic Regression (LR) | Low | Linear relationships | High |
| Support Vector Machine (SVM) | Medium | Non-linear relationships | Medium |
| Random Forest (RF) | High | Complex datasets, feature importance | Medium |
| Extreme Gradient Boosting (XGBoost) | High | Large datasets, predictive accuracy | Medium |
The applicability domain (AD) defines the structural space where the model can reliably predict activity, helping users identify when compounds fall outside the model's training set, which is crucial for regulatory acceptance [37].
QSAR Development Workflow and Methodologies
Data Acquisition and Curation
The foundation of any robust QSAR model is a high-quality dataset with well-defined endpoints. For environmental applications, key toxicity endpoints include:
- Aquatic toxicity: LC50 for fish, Daphnia, algae
- Bioaccumulation potential: Bioconcentration factor (BCF)
- Environmental persistence: Biodegradation half-life
- Environmental mobility: Soil adsorption coefficient (Koc)
Data sources for QSAR development include publicly available databases (EPA ECOTOX, REACH registration dossiers) and proprietary collections. Critical curation steps involve checking for duplicates, verifying experimental conditions, and standardizing measurement units [35]. For regulatory applications, data should comply with standardized testing guidelines (OECD, EPA) to ensure consistency and reliability.
Chemical Representation and Feature Generation
Molecular structure representation forms the basis for descriptor calculation. The process typically begins with structure representation (SMILES, InChI, or 2D/3D molecular files), followed by geometry optimization and descriptor computation using tools such as PaDEL, RDKit, or Dragon.
Table 2: Essential Descriptor Categories for Environmental QSAR
| Descriptor Category | Key Parameters | Environmental Relevance |
|---|---|---|
| Hydrophobic | log P, log D, water solubility | Bioaccumulation, membrane permeability |
| Electronic | pKa, HOMO/LUMO energies, polarizability | Reactivity, transformation potential |
| Steric | Molecular weight, molar volume, refractivity | Molecular transport, enzyme interactions |
| Topological | Connectivity indices, molecular fingerprints | Similarity assessment, read-across |
| Quantum Chemical | Partial charges, electrostatic potential | Reaction pathways, metabolite formation |
Feature selection techniques (genetic algorithms, stepwise regression, Random Forest importance) help identify the most relevant descriptors, reducing dimensionality and minimizing the risk of overfitting.
Model Building and Validation
The OECD principles for QSAR validation provide a framework for developing scientifically valid models, requiring: (1) a defined endpoint; (2) an unambiguous algorithm; (3) a defined domain of applicability; (4) appropriate measures of goodness-of-fit, robustness, and predictivity; and (5) a mechanistic interpretation, when possible [35].
Model validation employs several complementary approaches:
- Internal validation: Cross-validation techniques (k-fold, leave-one-out) assess robustness within the training set
- External validation: Prediction of a completely independent test set provides the gold standard for evaluating predictive ability
- Y-randomization: Verifies that models capture real structure-activity relationships rather than chance correlations
Performance metrics vary based on the endpoint type:
- Regression models: R², Q², RMSE, MAE
- Classification models: Accuracy, sensitivity, specificity, balanced accuracy, Matthews correlation coefficient
The following workflow diagram illustrates the comprehensive QSAR development process:
QSAR Applications in Environmental Risk Assessment
Tiered Approaches for Environmental Risk Assessment
Environmental risk assessment typically employs tiered approaches that begin with conservative screening models and progress to more sophisticated tools as needed. QSAR models effectively support initial tiers by prioritizing chemicals for further testing or identifying potentially hazardous compounds requiring regulatory attention [1] [39]. This approach balances resource allocation with protection goals, focusing experimental efforts on chemicals of highest concern.
In tiered ERA frameworks, QSAR applications include:
- Screening and prioritization: Identifying chemicals with potential persistence, bioaccumulation, or toxicity concerns
- Category formation: Grouping chemicals based on structural similarity for read-across
- Data gap filling: Providing predictions for untested compounds where experimental data are lacking
- Mixture assessment: Supporting evaluation of combined effects from multiple chemicals
Specific Environmental Application Domains
Persistence, Bioaccumulation, and Toxicity (PBT) Assessment
QSAR models play a crucial role in predicting the PBT profiles of chemicals, particularly for cosmetic ingredients where animal testing bans have created significant data gaps [37]. Comparative studies have identified high-performing models for key environmental parameters:
Table 3: High-Performing QSAR Models for Environmental Fate Parameters
| Environmental Parameter | High-Performing Models | Key Applications |
|---|---|---|
| Ready Biodegradability | Ready Biodegradability IRFMN (VEGA), Leadscope (Danish QSAR), BIOWIN (EPISUITE) | Persistence screening |
| Log Kow (Lipophilicity) | ALogP (VEGA), ADMETLab 3.0, KOWWIN (EPISUITE) | Bioaccumulation potential |
| Bioconcentration Factor (BCF) | Arnot-Gobas (VEGA), KNN-Read Across (VEGA) | Bioaccumulation assessment |
| Soil Adsorption (Koc) | OPERA v.2.0.0, KOCWIN (VEGA) | Environmental mobility |
These models support regulatory decisions under frameworks such as REACH and CLP, with qualitative predictions (e.g., biodegradable vs. non-biodegradable) generally proving more reliable than quantitative predictions when evaluated against regulatory criteria [37].
Pesticide Risk Assessment
Pesticides represent a particularly important application area for QSAR in ERA due to their intentional release into the environment and potential ecological impacts. Computational tools address both exposure assessment (predicting environmental concentrations) and effects assessment (predicting toxicity to non-target organisms) [2].
Exposure modeling tools predict pesticide distribution in environmental compartments:
- AGDISP: Predicts spray drift and deposition in air systems
- TOXSWA: Models pesticide fate in surface water systems
- PRZM: Simulates pesticide transport in the vadose zone
Toxicity prediction models estimate hazards to ecological receptors:
- BeeTox: Graph attention convolutional neural network model for predicting honeybee toxicity (accuracy: 0.837)
- EcoSAR: Predictive models for fish and aquatic invertebrate toxicity
- Plant Protection Product models: Species-specific toxicity predictions for regulatory submissions
The following diagram illustrates the pesticide risk assessment framework incorporating QSAR approaches:
Advanced QSAR Frameworks and Emerging Approaches
Knowledge-Enhanced QSAR Models
Traditional QSAR approaches based solely on chemical structure face limitations in predicting complex toxicological endpoints, particularly for pharmaceuticals where minor structural modifications can cause significant toxicity changes. The emerging paradigm of Quantitative Knowledge-Activity Relationships (QKARs) addresses these limitations by incorporating domain-specific knowledge alongside structural information [36].
QKAR development involves generating knowledge representations using large language models (e.g., GPT-4o) with specialized prompts that extract toxicologically relevant information, which is then converted to numerical vectors using text embedding models (e.g., text-embedding-3-large). These knowledge representations capture information beyond structural features, including:
- Drug class, indications, and usage patterns
- Metabolic pathways and toxic metabolites
- Mechanisms of toxicity
- Clinical case reports and regulatory actions
- Patient risk factors and biomarkers
In comparative studies, QKAR models consistently outperformed traditional QSAR approaches for predicting drug-induced liver injury (DILI) and drug-induced cardiotoxicity (DICT). Notably, QKARs demonstrated superior capability in differentiating drugs with similar structures but different toxicity profiles [36]. Hybrid approaches integrating knowledge-based and structure-based representations (designated Q(K + S)ARs) showed further enhanced prediction accuracy.
Visualization and Interpretation Tools
Model interpretation remains critical for regulatory acceptance and scientific understanding. Visual validation approaches, such as those implemented in CheS-Mapper 2.0, enable researchers to graphically inspect QSAR model validation results in three-dimensional chemical space [38]. This model-independent approach facilitates:
- Identification of misclassified compounds and potential data quality issues
- Assessment of whether models are overfitting or underfitting
- Detection of activity cliffs (structurally similar compounds with large activity differences)
- Mechanistic interpretation through chemical grouping
CheS-Mapper combines clustering, dimensionality reduction, and 3D visualization, representing each compound by its chemical structure rather than abstract symbols. This enables direct visual comparison of actual versus predicted activity values across chemical space, highlighting regions where models perform well or poorly.
Integrated Testing Strategies
Modern environmental risk assessment employs integrated approaches that combine QSAR predictions with experimental data and other in silico tools within weight-of-evidence frameworks [39]. These strategies acknowledge that no single method provides complete information but together can support robust decisions. Key integration approaches include:
- Read-across: Using data from similar compounds (source substances) to predict properties of a target substance
- Weight of evidence: Combining results from multiple QSAR models and other sources
- Toxicokinetic modeling: Integrating physiologically based kinetic models with QSAR predictions
- Adverse outcome pathways: Linking molecular initiating events to ecological outcomes through defined pathways
Table 4: Essential Resources for QSAR Development and Application
| Resource Category | Specific Tools | Primary Function |
|---|---|---|
| Descriptor Calculation | PaDEL, RDKit, Dragon, Mordred | Generate molecular descriptors from chemical structures |
| Model Development | Scikit-learn, Knime, Orange, Weka | Machine learning algorithms for QSAR building |
| Specialized QSAR Platforms | VEGA, EPISUITE, TEST, Danish QSAR | Integrated platforms with pre-built models |
| Validation & Visualization | CheS-Mapper 2.0, QSAR-Co | Model validation and visual analysis |
| Chemical Databases | EPA ECOTOX, PubChem, ChEMBL | Source of chemical structures and experimental data |
| Regulatory Support | OECD QSAR Toolbox, AMBIT | Read-across and category formation for regulatory compliance |
QSAR modeling represents a powerful approach for predicting chemical toxicity that continues to evolve through integration with advanced artificial intelligence methods and expanding biological knowledge. As regulatory frameworks increasingly emphasize New Approach Methodologies and seek to reduce animal testing, QSAR's role in environmental risk assessment will continue to expand. Future developments will likely focus on enhancing model interpretability, expanding applicability domains, and improving integration with adverse outcome pathways and systems toxicology approaches. The successful application of QSAR in regulatory contexts requires ongoing attention to validation, transparency, and defined applicability domains—ensuring that models provide reliable predictions for their intended purposes while clearly communicating limitations. Through continued refinement and appropriate application, QSAR modeling will remain an essential component of integrated testing strategies for environmental protection and chemical safety assessment.
Read-across is a methodology used to predict the properties of a data-poor target chemical by using experimental data from one or more structurally or biologically similar source compounds that are well-studied [40]. This approach represents a cornerstone of New Approach Methodologies (NAMs) in toxicology and environmental risk assessment, allowing researchers and regulators to fill critical data gaps without conducting new animal testing [41]. The fundamental premise of read-across is that chemicals with similar structural features may exhibit similar biological activities, environmental fate, and toxicological properties [40] [42].
Within the broader context of in silico environmental risk assessment research, read-across serves as a bridge between traditional animal studies and fully computational approaches. As regulatory agencies worldwide increasingly prioritize the reduction of animal testing while maintaining chemical safety standards, read-across has emerged as a scientifically valid and regulatory-accepted approach for hazard assessment [43] [41]. The methodology aligns with the goals of modern chemical management frameworks such as the European Union's REACH regulation and the U.S. EPA's Toxic Substances Control Act, which encourage the use of alternative methods for generating safety data [40] [43].
The application of read-across extends across multiple domains, including environmental risk assessment, pharmaceutical safety evaluation, and cosmetics safety [40] [24] [44]. For environmental risk assessment specifically, read-across helps predict the fate and effects of chemicals on ecosystems, supporting decisions about chemical registration, restriction, and remediation [2] [24]. This technical guide explores the principles, methodologies, and practical applications of read-across, with particular emphasis on its role within computational environmental risk assessment paradigms.
Fundamental Principles and Regulatory Context
Core Concepts and Definitions
Read-across operates on several key concepts that form the foundation of its application:
- Target Chemical: The data-poor substance for which predictions are needed [41]
- Source Chemical/Analogue: The well-characterized substance used to make predictions about the target [41]
- Chemical Category: A group of chemicals whose physicochemical, toxicological, and environmental fate properties are likely to be similar or follow a regular pattern due to structural similarity [40]
- Analogue Approach: Using one or a few source chemicals to predict properties of the target [41]
- Category Approach: Using a larger group of related chemicals where toxicological properties follow a predictable trend [41]
The scientific basis for read-across rests on the principle that chemical structure determines physical-chemical properties, which in turn determine environmental fate and biological effects [40]. This structure-activity relationship (SAR) foundation allows for extrapolation from data-rich to data-poor chemicals when sufficient similarity can be demonstrated.
Regulatory Framework and Acceptance
Read-across has gained significant traction in regulatory systems worldwide, though acceptance criteria and implementation frameworks vary:
Table: Regulatory Status of Read-Across in Different Jurisdictions
| Jurisdiction | Regulatory Framework | Status of Read-Across | Key Characteristics |
|---|---|---|---|
| European Union | REACH, EFSA | Formalized guidance | EFSA draft guidance (2025) outlines structured approach [41] |
| United States | TSCA, Superfund program | Case-by-case acceptance | Used in PPRTV assessments for data-poor chemicals [40] [42] |
| Republic of Korea | AREC (2019) | Allowed with limitations | Accepts alternative data including read-across [43] |
| International | ICH M7 Guideline | Accepted for pharmaceuticals | Used for establishing acceptable intake of impurities [43] |
Regulatory acceptance of read-across depends heavily on demonstrating sufficient similarity between source and target chemicals, with particular emphasis on the specific endpoint being assessed [43] [41]. The European Food Safety Authority (EFSA) has developed a structured approach that includes problem formulation, substance characterization, source identification, data gap filling, uncertainty assessment, and comprehensive reporting [41]. Similarly, the U.S. EPA has incorporated read-across into its Provisional Peer-Reviewed Toxicity Value (PPRTV) assessments for Superfund program chemicals that lack toxicity data [40].
A significant challenge in regulatory acceptance is the inconsistency in robustness of scientific evidence and lack of standardized acceptance criteria across agencies [43]. Regulatory bodies typically expect read-across to be supported not just by structural similarity but also by mechanistic evidence, such as mode of action or kinetic data, which can be difficult to obtain for data-poor chemicals [41].
Methodological Framework and Workflow
Tiered Approach to Read-Across Assessment
A systematic, tiered approach ensures rigorous implementation of read-across methodology. The following workflow visualization outlines the key stages in a comprehensive read-across assessment:
Workflow Title: Read-Across Assessment Process
Problem Formulation and Target Characterization
The initial stage involves clearly defining the assessment objectives and characterizing the target chemical:
- Define Assessment Goals: Identify the specific endpoints requiring prediction (e.g., acute aquatic toxicity, repeated dose toxicity) and the required level of certainty [40] [45]
- Characterize Target Chemical: Gather available data on physicochemical properties, structural features, and existing toxicological information [44]
- Identify Use Scenarios: Determine exposure contexts (environmental compartments, exposure routes, concentrations) that influence data needs [44]
Analogue Identification and Similarity Justification
Identifying suitable analogues requires a systematic approach to demonstrate similarity:
Table: Types of Similarity in Read-Across Justification
| Similarity Type | Description | Assessment Methods | Regulatory Importance |
|---|---|---|---|
| Structural Similarity | Shared functional groups, carbon chain length, molecular weight | Computational fingerprinting, expert judgment | Fundamental requirement; necessary but often insufficient alone [40] |
| Toxicokinetic Similarity | Similar absorption, distribution, metabolism, excretion | In vitro metabolism studies, PBPK modeling | Increasingly expected by regulators; strengthens justification [40] [44] |
| Toxicodynamic Similarity | Shared mode of action, target receptors, adverse outcome pathways | In vitro bioassays, molecular docking, toxicogenomics | Provides mechanistic support; highly valued in WoE assessment [40] [44] |
| Metabolic Similarity | Common metabolic pathways and transformation products | In silico metabolism prediction, experimental metabolite identification | Critical when target is metabolite of source chemical [40] |
The following diagram illustrates the category development and similarity justification process:
Diagram Title: Chemical Category Development Process
Experimental Protocols and Technical Approaches
Computational Tools for Analogue Identification
Several validated computational tools support the identification of suitable analogues and category formation:
- OECD QSAR Toolbox: Provides functionality for chemical categorization, trend analysis, and filling data gaps [41]
- EPA CompTox Chemicals Dashboard: Offers access to chemistry, toxicity, and exposure data for thousands of chemicals [40] [41]
- CEFIC AMBIT: Open-source software for chemical grouping and read-across [41]
- EPA Analog Identification Methodology (AIM) Tool: Specifically designed to identify structural analogues [41]
These tools employ various chemical fingerprinting algorithms and similarity metrics to quantify structural relationships between chemicals. The most common approaches include Tanimoto similarity, maximum common substructure, and functional group analysis.
New Approach Methodologies (NAMs) for Similarity Assessment
Integrating data from New Approach Methodologies strengthens read-across hypotheses by providing mechanistic evidence:
In Vitro Toxicokinetics Assessment
Protocol: Hepatic Metabolism Studies Using Human Hepatocytes
- Objective: Compare metabolic pathways between source and target chemicals
- Materials: Cryopreserved human hepatocytes, Williams E medium, test chemicals
- Procedure:
- Thaw hepatocytes and seed in collagen-coated plates (1×10^6 viable cells/well)
- Incubate with test chemicals (1-100 μM) for 0-24 hours
- Collect samples at multiple time points (0, 0.5, 1, 2, 4, 8, 24 hours)
- Analyze parent compound depletion and metabolite formation using LC-MS/MS
- Compare metabolic profiles and half-lives between source and target chemicals
- Data Interpretation: Similar metabolic profiles and comparable half-lives support kinetic similarity [44]
In Vitro Toxicodynamics Assessment
Protocol: High-Throughput Transcriptomics in RTgill-W1 Cells
- Objective: Compare gene expression profiles induced by source and target chemicals
- Materials: RTgill-W1 cell line, cell culture reagents, RNA extraction kit, microarray or RNA-seq platform
- Procedure:
- Culture RTgill-W1 cells according to OECD TG 249 in 96-well plates
- Expose cells to a range of concentrations (0.1-100 μM) for 24 hours
- Extract total RNA and assess quality (RIN >7.0)
- Perform gene expression analysis using targeted RNA-seq or microarray
- Identify differentially expressed genes (fold-change >2, p-value <0.05)
- Compare enriched pathways and gene signatures between chemicals
- Data Interpretation: Shared pathway perturbations support toxicodynamic similarity [9]
Quantitative Read-Across and Point of Departure Translation
For quantitative risk assessment, read-across involves transferring points of departure (PODs) from source to target chemicals:
Protocol: Physiologically Based Pharmacokinetic (PBPK) Modeling for Dose Translation
- Objective: Extrapolate in vitro PODs to human relevant exposures
- Materials: PBPK modeling software, in vitro to in vivo extrapolation parameters, chemical-specific ADME data
- Procedure:
- Develop or adapt existing PBPK models for source and target chemicals
- Incorporate dermal absorption parameters from in vitro skin penetration studies
- Calibrate models using available in vivo pharmacokinetic data
- Convert in vitro PODs to bioequivalent human doses using reverse dosimetry
- Apply appropriate uncertainty factors for inter-species and inter-individual variability
- Data Interpretation: PBPK modeling provides quantitative bridge between in vitro effects and human exposure scenarios [44]
Case Studies and Practical Applications
Case Study 1: Phosphoramide Compounds (U.S. EPA Superfund Program)
The U.S. EPA applied read-across to derive screening-level toxicity values for pentamethylphosphoramide (PMPA) and tetramethylphosphoramide (TMPA) using hexamethylphosphoramide (HMPA) as a source analogue [40].
- Structural Similarity: All compounds contain phosphoric acid amine derivatives with nitrogen-phosphorous bonds
- Metabolic Relationship: PMPA and TMPA are primary intermediate metabolites of HMPA via sequential demethylation by CYP450 enzymes
- Toxicological Similarity: All compounds cause degenerative nasal lesions in rats due to preferential deposition and metabolism in nasal tissue
- Regulatory Application: HMPA's point of departure (15 mg/kg-day based on nasal lesions) was adopted for PMPA and TMPA, enabling Superfund site risk assessments [40]
Case Study 2: Isoflavones in Cosmetics (Next-Generation Risk Assessment)
A comprehensive read-across case study established safe use levels of daidzein in cosmetic products using genistein as an analogue [44].
- Assessment Framework: 10-step tiered approach using exclusively NAMs
- Key Endpoints: Endocrine disruption potential via ERα assay (most sensitive endpoint)
- Methodologies: PBPK modeling, in vitro biokinetics, toxicogenomics, EATS assays
- Outcome: Determined safe concentration of 0.1% daidzein in body lotion without animal testing
- Regulatory Significance: Demonstrated proof-of-concept for NAM-based safety assessments in cosmetics [44]
Case Study 3: Pesticide Environmental Risk Assessment
Read-across has been successfully applied to predict environmental fate and effects of pesticides:
- Application: Predicting acute toxicity to honeybees using structural analogues
- Tools: BeeTox model using graph attention convolutional neural networks
- Performance: Achieved prediction accuracy of 0.837, specificity of 0.891, and sensitivity of 0.698
- Regulatory Impact: Reduced need for animal testing while maintaining protective risk assessments [2]
Successful implementation of read-across requires access to specific tools, databases, and methodological resources:
Table: Essential Research Resources for Read-Across Assessments
| Resource Category | Specific Tools/Databases | Function/Purpose | Access |
|---|---|---|---|
| Chemical Databases | EPA CompTox Dashboard, eChemPortal | Access to chemical structures, properties, and toxicity data | Public [40] [41] |
| Grouping Tools | OECD QSAR Toolbox, AIM Tool, AMBIT | Identify structural analogues and form chemical categories | Public/Commercial [41] |
| Bioactivity Data | ToxCast, Tox21 | High-throughput screening data for mechanistic similarity | Public [41] [9] |
| Metabolism Prediction | GLORYx, Meteor Nexus | Predict metabolic pathways and transformation products | Public/Commercial [44] |
| PBPK Modeling | Generic PBPK platforms, Open-source tools | Extrapolate in vitro data to in vivo exposures | Various [44] |
| Toxicogenomics | LINCS, Connectivity Map | Compare gene expression signatures | Public [9] |
Uncertainty Analysis and Limitations
Despite its utility, read-across carries inherent uncertainties that must be transparently addressed:
- Structural Discontinuity: Small structural differences may lead to significant toxicological differences [41]
- Mechanistic Understanding: Limited knowledge of mode of action can undermine similarity justifications [45]
- Metabolic Variations: Species-specific metabolism may not be adequately captured [40]
- Data Quality: Inconsistent data quality for source chemicals affects reliability of predictions [43]
The weight of evidence approach systematically addresses these uncertainties by integrating multiple lines of evidence [40] [45]. The U.S. EPA and EFSA frameworks emphasize transparent documentation of uncertainty sources and application of appropriate assessment factors to ensure protective outcomes [40] [41].
Read-across methodology continues to evolve with advancements in computational toxicology and systems biology. Future developments will likely focus on:
- Integration of High-Content NAMs: Increased use of transcriptomics, epigenomics, and high-throughput phenotypic profiling [9] [44]
- Artificial Intelligence Applications: Machine learning and deep learning algorithms for similarity quantification and prediction [2]
- Quantitative Uncertainty Frameworks: Standardized approaches for quantifying and reporting read-across uncertainty [45]
- Regulatory Harmonization: International convergence on read-across acceptance criteria and reporting standards [43]
In conclusion, read-across represents a scientifically robust and regulatory accepted methodology for filling data gaps in chemical risk assessment. When implemented within a systematic framework that integrates structural, toxicokinetic, and toxicodynamic similarity, supported by New Approach Methodologies, read-across enables protective decision-making while reducing reliance on animal testing. As computational capabilities advance and biological understanding deepens, read-across will play an increasingly central role in next-generation environmental risk assessment paradigms.
In silico environmental risk assessment (ERA) represents a paradigm shift from traditional, empirical toxicity testing toward a mechanistic, computational approach. This transition is driven by the need to evaluate chemical safety more efficiently, reduce animal testing, and understand the effects of chemicals under realistic, time-variable exposure scenarios [28] [46]. At the forefront of this shift are toxicokinetic-toxicodynamic (TK-TD) models, which mathematically describe the processes of chemical uptake, distribution, metabolism, and the subsequent toxic effects on organisms [47]. A particularly powerful class of these models is based on Dynamic Energy Budget (DEB) theory, which provides a physiological framework for understanding how organisms acquire and utilize energy, and how chemical stressors disrupt these fundamental processes [48].
These biologically-based models are core components of New Approach Methodologies (NAMs), which aim to modernize risk assessment by leveraging in vitro and in silico tools [28] [46]. Their application is critical for addressing the "zero pollution" ambition and the Safe-and-Sustainable-by-Design (SSbD) framework promoted by European Union policies [28]. By simulating the essential biological processes of energy allocation and toxicant action, TK-TD and DEB models enable extrapolations from laboratory data to complex field conditions, providing a scientifically robust foundation for ecological and human health protection.
Theoretical Foundations of TK-TD and DEB Models
Core Components of Toxicokinetic-Toxicodynamic (TK-TD) Models
TK-TD models dissect the action of a toxicant into two sequential processes [47]:
- Toxicokinetics (TK): Describes the time-course of a chemical within an organism, answering "what the body does to the chemical." This includes uptake, distribution, metabolism, and excretion (ADME processes). The fundamental TK equation for the scaled internal concentration, ( Dw(t) ), is often modeled as:
\[ \frac{d{D}_{w}(t)}{dt}={k}_{d}({C}_{w}(t)-{D}_{w}(t)) \]where ( Cw(t) ) is the time-varying external concentration, and ( k_d ) is the dominant rate constant representing the slowest process dominating toxicity dynamics [47]. - Toxicodynamics (TD): Describes the biochemical and physiological mechanisms of toxic action, answering "what the chemical does to the body." It quantifies the damage and resulting adverse effects once the toxicant reaches its target site. For survival, the General Unified Threshold Model of Survival (GUTS) integrates TK with two main TD concepts [47]:
- GUTS-SD (Stochastic Death): Assumes all individuals have an identical internal threshold concentration (( z )); death is a stochastic process once this threshold is exceeded.
- GUTS-IT (Individual Tolerance): Assumes individual thresholds vary in the population according to a distribution (e.g., log-logistic); an individual dies immediately when its threshold is exceeded.
Principles of Dynamic Energy Budget (DEB) Theory
DEB theory provides a quantitative, mechanistic framework for understanding the energy metabolism of an organism throughout its life cycle [48]. Its core principle is the conserved allocation of energy: energy assimilated from food is allocated to various life-supporting processes following fixed rules, prioritizing maintenance, then growth, development, and finally reproduction [49] [48]. In ecotoxicology, DEB theory is extended with TK-TD modules to create DEB-TKTD models. These models simulate how a chemical stressor interferes with the energy budget, leading to observable effects on growth, reproduction, and survival [49]. The physiological Mode of Action (pMoA) defines how the toxicant stressor affects energy allocation, for example, by increasing maintenance costs, reducing assimilation, or directly hampering reproduction [49].
The AOP-DEB Model Linkage
A key advancement is the conceptual linkage between Adverse Outcome Pathways (AOPs) and DEB models [50]. AOPs provide a "bottom-up" description of a sequence of biologically measurable events, from a Molecular Initiating Event (MIE) to an Adverse Outcome (AO) at the organism level. DEB models offer a "top-down" framework that reverse-engineers stressor effects on growth, reproduction, and survival into changes in energy allocation. The two approaches are complementary; key events in an AOP can be interpreted as measures of damage-inducing processes that affect specific DEB variables or rates, thereby providing a mechanistic bridge from suborganismal disruption to population-level consequences [50].
Model Implementation and Experimental Protocols
Calibration and Validation of TK-TD Models
Implementing TK-TD models requires a rigorous process of calibration and validation using laboratory toxicity data [51]. The workflow involves distinct, interconnected phases as shown in the diagram below.
Detailed Experimental Protocol for Model Calibration:
- Standard Laboratory Toxicity Tests: Conduct tests following standardized guidelines (e.g., OECD Test Guideline 211 for Ceriodaphnia dubia reproduction) [51]. The design should include:
- A constant concentration experiment with multiple concentrations and a control, measuring survival, growth, and reproduction over a significant part of the organism's life cycle (e.g., 21 days for D. magna) [49].
- A time-variable exposure experiment with pulsed exposures of different heights and frequencies to inform the model on TK processes under non-steady-state conditions [47] [51].
- Model Calibration: Use the data from the constant exposure test to estimate model parameters.
- TK parameters (e.g., uptake rate ( k_d ), elimination rate) are inferred from the time-course of effects or direct body residue measurements if available.
- TD parameters are estimated by fitting the model to the observed survival, growth, and reproduction data. This includes the No-Effect Concentration (NEC), the killing rate, and parameters related to the pMoA [47] [49].
- A Bayesian framework is recommended for calibration as it allows for robust characterization of parameter uncertainties and their propagation to model predictions, such as LC(x, t) and multiplication factors (MF(x, t)) [47].
- Model Validation: Design an independent validation experiment with an untested exposure profile (e.g., a series of pulses with different intervals) [51]. Compare the model predictions against the observed outcomes without further adjusting the calibrated parameters. This step is crucial for establishing model credibility for regulatory use [51].
- Environmental Prediction: Apply the validated model to realistic environmental exposure profiles, such as those generated by the FOCUS (Forum for Co-ordination of Pesticide Fate Models and Their Use) framework for pesticides in surface water [51] [49]. A moving time window approach is used to assess risk across the entire exposure profile, reducing uncertainty compared to static methods [51].
Case Study: TK-TD Modeling of Nanoplastics Toxicity
A recent study demonstrated the application of a TK-TD framework to assess the ecological risks of differently sized Polystyrene Nanoplastics (PS NPs) on Daphnia magna [52].
- Objective: To elucidate how particle size (30, 60, 80, 120, and 200 nm) influences the bioaccumulation and toxicity of PS NPs and to establish environmental safety thresholds.
- Methods:
- TK Modeling: AIE (Aggregation-Induced Emission) fluorogen-labeled PS NPs were used to precisely track uptake and elimination in D. magna. Uptake and elimination rate constants were quantified for each particle size.
- TD Modeling: Acute toxicity tests (survival) were conducted for each PS NP size. The GUTS model was calibrated to this data to derive size-specific TD parameters.
- Mechanistic Insight: Transcriptomic analysis was performed to identify disrupted pathways (e.g., digestion and oxidative stress).
- Findings: The study found smaller PS NPs (e.g., PS30) were more toxic than larger ones, primarily accumulating in thoracic appendages and disrupting nutrient digestion. The TK-TD model was used to predict 48-h LC50 and No-Effect Concentrations (NECs) for each particle size, providing a foundational dataset for environmental threshold derivation [52].
Table 1: Key Parameters from TK-TD Modeling of Polystyrene Nanoplastics in Daphnia magnacitation:8
| Particle Size (nm) | Uptake Rate (L g⁻¹ day⁻¹) | Elimination Rate (day⁻¹) | 48-h LC50 (mg L⁻¹) | No-Effect Concentration (NEC) (mg L⁻¹) |
|---|---|---|---|---|
| PS30 | 1.21 | 0.56 | 1.2 | 0.15 |
| PS60 | 1.45 | 0.61 | 3.0 | 1.05 |
| PS80 | 1.58 | 0.65 | 14.4 | 8.2 |
| PS120 | 1.38 | 0.48 | 13.5 | 6.9 |
| PS200 | 1.89 | 0.72 | 196.7 | 92.7 |
Comparative Analysis of DEB Models
DEBtox2019 vs. Standard DEB-TKTD Model
The implementation of DEB theory in ecotoxicology has led to models of varying complexity. A 2024 study directly compared two prevalent DEB-TKTD models [53] [49]:
- DEBtox2019: A simplified model based on the DEBkiss framework, using compound parameters with direct links to observations (e.g., maximum body length).
- Standard DEB-TKTD (stdDEB-TKTD): A more complex model based on the full standard DEB animal model, using primary parameters with direct links to underlying metabolic processes (e.g., volume-specific maintenance costs).
Table 2: Comparison of DEB-TKTD Models for Ecological Risk Assessment [53] [49]
| Feature | DEBtox2019 (Simplified) | Standard DEB-TKTD (Complex) |
|---|---|---|
| Model Basis | DEBkiss framework | Standard DEB animal model |
| Parameters | Compound parameters (e.g., maximum body length) | Primary parameters (e.g., volume-specific maintenance costs) |
| Reserve Compartment | Absent (direct use of assimilated energy) | Present (explicit energy buffering) |
| Maturation Tracking | Via body-size thresholds | Via maturity level as a state variable |
| Calibration Data | Can be parameterized with data from an ecotoxicity test | Requires a pre-existing species-specific parameter set (e.g., AmP) |
| Ease of Use | Higher (fewer parameters, direct link to data) | Lower (requires deeper DEB theory knowledge) |
| Model Flexibility | Lower (tailored for standard test species and endpoints) | Higher (applicable to a wider range of species and contexts) |
| Performance (Fit & Prediction) | Similar to stdDEB-TKTD after harmonization of modeling choices | Similar to DEBtox2019 after harmonization of modeling choices |
The critical finding of the comparison was that after harmonizing modeling choices (e.g., treatment of starvation, damage dynamics), both models achieved very similar performance in both calibration to laboratory data and forward prediction of effects under time-variable exposure profiles [49]. Consequently, model selection for ERA cannot be based on goodness-of-fit alone but should consider the trade-off between ease of use (favoring DEBtox2019) and model flexibility (favoring stdDEB-TKTD) for addressing complex biological questions [49].
Successful implementation of TK-TD and DEB modeling relies on a suite of computational and biological resources.
Table 3: Key Research Reagent Solutions for TK-TD and DEB Modeling
| Resource Name | Type | Function in Research | Example / Source |
|---|---|---|---|
| BYOM Platform | Software Platform | A flexible, open-source model platform for coding, calibrating, and analyzing TK-TD models. | DEBtox.info (BYOM, Ver. 4.5) [51] |
| Add-my-Pet (AmP) Database | Biological Database | A curated library of life-cycle data and DEB parameters for over 857 species. | AmP Database [48] |
| GUTS Model (in morse R package) | Software Package | An R package implementing the General Unified Threshold Model of Survival (GUTS) for survival analysis. | morse package [47] |
| AIE Fluorogen-Labeled NPs | Research Reagent | Enables precise tracking of nanomaterial uptake and distribution in organisms without dye leakage. | TPA-labeled Polystyrene NPs [52] |
| FOCUS Exposure Profiles | Data & Methodology | Standardized, time-variable environmental exposure scenarios for pesticides in surface water. | FOCUS Surface Water Scenarios [51] [49] |
TK-TD and DEB models represent a cornerstone of modern, in silico environmental risk assessment. By mechanistically linking exposure to internal concentrations and subsequent toxic effects within a framework of fundamental energy allocation principles, these models provide a powerful tool for extrapolating laboratory results to realistic environmental scenarios [51] [48]. The integration of these models with AOPs enhances their mechanistic credibility, while the development of comprehensive databases and user-friendly software platforms increases their accessibility for regulators and researchers [46] [50].
As the field progresses, the application of these models is expanding beyond traditional chemical risk assessment to include novel stressors like nanoplastics [52] and is being explored in occupational health contexts under the Next Generation Risk Assessment (NGRA) paradigm [28]. The ongoing challenge lies in the standardization of modeling practices, managing uncertainty, and fostering broader regulatory acceptance. However, the demonstrated ability of TK-TD and DEB models to integrate diverse data, reduce uncertainty, and support predictive, hypothesis-driven risk assessment ensures their enduring role in the development of a safe and sustainable chemical economy.
In silico exposure modeling represents a transformative approach in environmental risk assessment (ERA), using computational tools to predict the fate and transport of chemicals in the environment. These models have become indispensable for evaluating chemical safety while reducing reliance on costly and time-consuming experimental studies. The framework for ERA of pesticides and other chemicals typically comprises four distinct steps: hazard identification, exposure assessment, toxicity assessment, and risk characterization [2]. Exposure modeling specifically addresses the critical question of how chemicals move and distribute through environmental compartments after release, enabling scientists to estimate concentrations that organisms and humans may encounter.
The advantages of in silico methods are substantial, offering rapid, cost-effective, and accurate alternatives to traditional testing approaches. Conventional pesticide testing can cost up to $9,919,000 overall, with chronic toxicity studies in animals taking up to two years to complete [2]. In silico approaches can potentially eliminate the use of 0.1–0.15 million test animals and save 50–70 billion US dollars when applied to 261 compounds [2]. For regulatory agencies like the Environmental Protection Agency (EPA), these tools provide essential capabilities for assessing chemicals under statutes such as the Toxic Substances Control Act (TSCA), particularly when laboratory studies or monitoring data are unavailable or need supplementation [54] [55].
Fundamental Principles of Chemical Fate and Transport
Key Processes Governing Chemical Behavior
Chemical fate in the environment is governed by interconnected processes that determine distribution, persistence, and ultimate sinks. When chemicals are introduced into the environment, they undergo complex transport and transformation processes across air, water, and soil compartments. Volatilization moves chemicals from soil and water surfaces into the atmosphere, while deposition returns them to terrestrial and aquatic systems. Degradation occurs through multiple pathways including hydrolysis in water and soil, photolysis in air and surface waters, and biodegradation mediated by microorganisms [2].
Spray application of pesticides demonstrates these complex pathways, with approximately 30% of applied pesticide potentially entering the atmosphere through spray drift and volatilization from soil and crops [2]. These chemicals then distribute across environmental media based on their physicochemical properties and environmental conditions. Persistent and Mobile (PM) organic chemicals represent a particular concern due to their ability to infiltrate natural barriers and contaminate drinking water sources, with their multimedia transport and distribution influenced by both the emission mode and local hydrology [56].
Critical Chemical Properties for Fate Prediction
The behavior of chemicals in environmental systems is largely determined by fundamental physicochemical properties that govern partitioning, persistence, and transport potential. Key properties include water solubility, which influences aquatic mobility; vapor pressure, which controls volatilization potential; and soil adsorption coefficients, which determine soil-water partitioning. Transformation rates such as hydrolysis, photolysis, and biodegradation half-lives indicate environmental persistence, while octanol-water partition coefficients (Kow) help predict bioaccumulation potential [54] [5].
Table 1: Essential Physicochemical Properties for Environmental Fate Prediction
| Property | Environmental Significance | Common Predictive Tools |
|---|---|---|
| Water Solubility | Determines aquatic mobility and bioavailability | EPI Suite, OPERA |
| Vapor Pressure | Controls volatilization potential | EPI Suite, QSAR Toolbox |
| Soil Adsorption Coefficient (Koc) | Predicts soil-water partitioning and groundwater contamination risk | EPI Suite, QSAR-ME Profiler |
| Octanol-Water Partition Coefficient (Kow) | Indicates bioaccumulation potential | EPI Suite, OPERA |
| Biodegradation Half-life | Measures environmental persistence | EPI Suite, University of Minnesota Biodegradation Database |
The identification of Persistent, Mobile and Toxic (PMT) and very Persistent and very Mobile (vPvM) substances has gained significant regulatory attention, particularly in the European Union where these categories have been incorporated into the Classification, Labeling, and Packaging (CLP) regulation EU 2023/707 [5]. These substances pose particular challenges for drinking water quality due to their ability to pass through natural and artificial barriers.
Computational Methodologies and Tools
Regulatory Modeling Approaches
Regulatory modeling frameworks provide standardized methodologies for chemical exposure assessment. The EPA employs a tiered approach where initial screening models use conservative assumptions to identify chemicals of concern, while higher-tiered tools incorporate more realistic parameters for refined assessments [54]. The Chemical Screening Tool for Exposures and Environmental Releases (ChemSTEER) estimates environmental releases and worker exposures resulting from chemical manufacture, processing, and use in industrial and commercial workplaces [54].
The Estimation Programs Interface (EPI Suite) represents a cornerstone of predictive fate assessment, providing estimates of physical/chemical properties and environmental fate parameters that indicate where a chemical will distribute in the environment and how long it will persist [54] [5]. For aquatic systems, the Point Source Calculator estimates chemical concentrations in water column, porewater, and sediment from point source discharges, while the upcoming ReachScan model will extend these capabilities to stream networks downstream from industrial facilities [54].
Spatial and Multimedia Modeling
Spatially distributed modeling approaches incorporate geographical variability in environmental parameters, moving beyond generic scenarios to account for specific regional characteristics. The refined PROTEX (PROduction-To-EXposure) model exemplifies this approach, enabling evaluation of environmental fate and human exposure to PM organic chemicals in different drinking water sources [56]. This model demonstrates how emission mode and drinking water source significantly influence exposure scenarios, with regionally released perfluorooctanoic acid (PFOA) predominantly accumulating in estuarine waters in humid regions while concentrating in deep groundwater in arid regions [56].
The AGricultural DISPersal model (AGDISP) has been successfully employed to monitor pesticide deposition and spray drift, effectively tracking atrazine drift up to 400 meters from application sites on sorghum fields [2]. Such tools are particularly valuable for assessing off-target movement and potential impacts on non-target organisms and ecosystems.
Machine Learning and Emerging Approaches
Machine learning (ML) is rapidly reshaping chemical exposure assessment, with bibliometric analyses revealing an exponential publication surge from 2015 onward, dominated by environmental science journals with China and the United States leading in research output [57]. Algorithm development has centered on XGBoost and random forests, with applications spanning water quality prediction, quantitative structure-activity relationship (QSAR) modeling, and specific contaminant classes like per- and polyfluoroalkyl substances (PFAS) [57].
The BeeTox model exemplifies advanced ML applications, employing graph attention convolutional neural networks (GACNN) to distinguish bee-toxic chemicals with prediction accuracy, specificity, and sensitivity of 0.837, 0.891, and 0.698, respectively [2]. Principal component analysis (PCA) further enhances these models by distinguishing pesticides with high potential toxicity to honeybees. These data-driven approaches require sizable datasets and effective molecular descriptors due to the intricate physicochemical and structural properties of chemicals, complex organism systems, agricultural practices, and varying climatic conditions [2].
Experimental Protocols and Methodologies
Protocol for Screening-Level Exposure Assessment
A systematic approach to screening-level exposure assessment integrates both computational and experimental data. The initial step involves compiling existing measured or monitoring data relevant to the assessment purpose [54]. For chemical characterization, researchers should gather or predict fundamental properties including water solubility, vapor pressure, soil adsorption coefficient (Koc), octanol-water partition coefficient (Kow), and degradation half-lives using tools such as EPI Suite or the OECD QSAR Toolbox [54] [5].
The exposure scenario definition must specify release patterns (continuous, intermittent, or single event), environmental media of initial release, and potential receptor populations (aquatic organisms, terrestrial species, or humans) [54]. For screening assessments, models like EPA's Exposure and Fate Assessment Screening Tool (E-FAST) provide conservative estimates using default parameters representing reasonable worst-case conditions [54]. Results should include estimated concentrations in relevant environmental media (air, water, soil, sediment) and comparison to toxicity thresholds for initial risk identification.
Protocol for Higher-Tiered Refined Assessment
Higher-tiered assessments replace conservative assumptions with realistic parameters to generate more precise exposure estimates. For spatial modeling, this involves incorporating geographically-specific data on soil types, hydrology, climate patterns, and land use [58]. For veterinary medicines and human pharmaceuticals, this includes modeling transport from manure-amended fields or wastewater treatment plants, considering transformation products and sorption dynamics [58].
Chemical-specific degradation data from laboratory studies should replace default rates, with attention to metabolite formation and potential toxicological significance. For mobile and persistent chemicals, the PROTEX model methodology can be applied to characterize multimedia distribution and potential for drinking water contamination, particularly for scenarios involving groundwater sources or arid regions where contamination may be significantly less reversible [56]. Model validation against available monitoring data strengthens confidence in predictions, with uncertainty analysis quantifying variability and informing risk management decisions.
Table 2: Computational Tools for Environmental Exposure Assessment
| Tool Name | Primary Application | Key Features | Regulatory Acceptance |
|---|---|---|---|
| EPI Suite | Physical/chemical properties and environmental fate prediction | Estimates degradation rates, distribution, and persistence | EPA recommended [54] |
| AGDISP | Pesticide spray drift prediction | Models atmospheric deposition and drift from application sites | Used in pesticide registration [2] |
| ChemSTEER | Industrial release and occupational exposure | Estimates workplace exposures and environmental releases | EPA TSCA assessments [54] |
| E-FAST | Screening-level exposure assessment | Models consumer, general public and environmental exposures | EPA TSCA assessments [54] |
| PROTEX | Multimedia fate of persistent, mobile chemicals | Tracks chemicals across environmental compartments including groundwater | Research application [56] |
| BeeTox | Ecological toxicity prediction | Graph neural network for bee toxicity classification | Preclinical screening [2] |
Applications in Chemical Risk Assessment
Pharmaceutical and Personal Care Products (PPCPs)
PPCPs represent emerging contaminants of concern due to their continuous release and potential biological effects at low concentrations. In silico hazard screening combining multiple QSAR tools (OECD Toolbox, OPERA, EPI Suite, QSAR-ME Profiler) has been applied to 245 PPCPs to identify persistent, mobile and toxic (PMT) substances [5]. This integrated approach successfully prioritized 16 compounds as most hazardous to the aquatic environment, with six associated with potential risk based on reported environmental concentrations [5].
Removal efficiency (RE) in wastewater treatment plants serves as a critical parameter for exposure assessment of PPCPs. Studies have characterized RE values across different treatment technologies, from conventional activated sludge systems to advanced tertiary treatments, providing essential data for predicting environmental loads [5]. The combination of experimental RE data with in silico predictions for persistence, bioaccumulation, and toxicity enables comprehensive risk-based prioritization of substances requiring regulatory attention or treatment optimization.
Pesticide Risk Assessment
Pesticides represent a major application area for exposure modeling due to their intentional release into the environment and potential impacts on non-target organisms. The AGDISP model has been extensively applied to predict spray drift, a significant pathway for off-target movement and potential ecological effects [2]. For soil and water compartments, models like TOXSWA (not detailed in search results) simulate pesticide fate in water-sediment systems, with field tests demonstrating reasonable agreement between simulated and observed chlorpyrifos concentrations in water, sediment, and macrophytes in stagnant ditches [2].
The integration of exposure modeling with toxicity assessment represents a critical advancement for pesticide risk characterization. The U.S. Environmental Protection Agency has reported that conventional pesticide testing can cost nearly $10 million overall, creating strong impetus for in silico approaches that can reduce animal testing while providing robust safety assessments [2].
Essential Software and Platforms
The computational toxicology landscape offers diverse tools for exposure and risk assessment. The Aggregated Computational Toxicology Resource (ACToR) serves as EPA's online warehouse of publicly available chemical toxicity data, aggregating information from over 1,000 public sources on more than 500,000 environmental chemicals [54]. The OECD eChemPortal provides complementary access to physical & chemical properties, ecotoxicity, and environmental fate data from international sources [54].
For biodegradation pathway prediction, the University of Minnesota Biocatalysis/Biodegradation Database includes a Pathway Prediction System (PPS) that predicts plausible pathways for microbial degradation of chemical compounds [54]. Specialized platforms like ToxStudio address specific safety assessment needs, incorporating AI-powered drug-induced liver injury prediction (Libra), cardiac safety simulation, and secondary pharmacology assessment [59].
Model Development and Validation Considerations
Credible evaluation systems require appropriate data and robust model validation. Research needs include addressing the accuracy and applicability of presented models, particularly when training datasets are limited [2]. For ML approaches, expanding chemical space coverage remains a priority, as current applications show biases toward certain chemical classes while underrepresenting others such as lignin, arsenic, and phthalates despite their fast-growing detection [57].
The migration of ML tools toward dose-response and regulatory applications necessitates greater attention to model interpretability and uncertainty quantification [57]. Explainable artificial intelligence (XAI) workflows represent an emerging frontier to enhance regulatory acceptance and practical implementation of complex ML models in chemical risk assessment [57].
In silico exposure modeling has evolved from a supplementary approach to a central methodology for predicting chemical fate in air, water, and soil. The integration of mechanistic models with machine learning approaches provides powerful capabilities for chemical safety assessment across regulatory, research, and industrial contexts. As chemical production continues to expand globally, these tools will play an increasingly critical role in prioritizing substances for further testing, identifying potential exposure hotspots, and supporting sustainable chemical design.
The successful application of computational tools for risk assessment depends on appropriate model selection, understanding of underlying assumptions, and transparent communication of uncertainties. Future developments will likely focus on enhancing model interoperability, expanding chemical space coverage, and strengthening the integration of exposure predictions with adverse outcome pathways for more holistic risk assessment. As regulatory agencies like the FDA advance policies to reduce animal testing, in silico exposure modeling will become increasingly essential for ensuring chemical safety while embracing New Approach Methodologies (NAMs) in environmental risk assessment.
In silico environmental risk assessment represents a paradigm shift in ecotoxicology, leveraging computational models to predict the hazards and exposures of chemicals, thereby reducing reliance on time-consuming and costly animal testing [60]. This approach is particularly vital for data-poor scenarios, such as evaluating new chemicals or, as explored in this case study, assessing the ecological risk of bisphenol A (BPA) alternatives. With BPA production facing increasing global restrictions, substitutes like bisphenol S (BPS) and bisphenol F (BPF) have been introduced. However, their close structural similarity to BPA raises concerns about potential similar ecological and health toxicity effects [60]. This case study details a methodology for using coupled in silico toxicology models to fulfill the core components of ecological risk assessment: hazard identification and characterization, exposure assessment, and risk characterization for these emerging contaminants.
Methodology: A Coupled In Silico Workflow
The assessment employs an integrated framework combining quantitative structure-activity relationship (QSAR) models and interspecies correlation estimation (ICE) models to generate sufficient toxicity data for constructing species sensitivity distributions (SSDs), which are crucial for deriving predicted no-effect concentrations (PNECs) [60].
Model Coupling and Workflow
The strength of this approach lies in the synergy between different in silico models. QSAR models predict toxicity based on chemical structure, filling data gaps where experimental data is unavailable. Subsequently, ICE models use available toxicity data from surrogate species to extrapolate toxicity to a wider range of untested species, thus expanding the dataset for SSD construction [60]. The following workflow diagram illustrates this integrated process.
Detailed Experimental Protocols
QSAR Model Application
- Platform Selection: The free, widely accepted VEGA platform (available at https://www.vegahub.eu) is used for QSAR predictions [60]. This software provides multiple QSAR models to predict toxicity properties for various endpoints.
- Input Data: The chemical structures of BPA, BPS, and BPF, along with their key physicochemical properties (e.g., Log Kow, molecular weight), serve as the primary input. These properties can be sourced from publicly accessible chemical databases such as the EPA CompTox Chemicals Dashboard, PubChem, and ChemSpider [60].
- Execution: The chemical structures are input into the platform, and relevant ecotoxicity models (e.g., for Daphnia magna, Pimephales promelas) are selected to predict chronic toxicity values. Each model within VEGA provides an assessment of its own reliability, which must be evaluated for each prediction [60].
ICE Model Application
- Platform Selection: The USEPA's Web-ICE application (available at https://www.epa.gov/web-ice) is used for interspecies extrapolation [60].
- Procedure: For a given chemical, available experimental or QSAR-predicted toxicity data for a "surrogate species" (e.g., Daphnia magna) is entered into the Web-ICE tool. The model then generates predictions of toxicity for multiple "predicted taxa" (e.g., fish, algae, other invertebrates) based on established statistical correlations between species [60].
- Data Expansion: This step is critical for generating the minimum sample size (typically 5 to 10 chronic toxicity values) required for robust SSD modeling [60].
Species Sensitivity Distribution (SSD) and PNEC Derivation
- SSD Construction: Chronic toxicity data (e.g., LC50, EC50) for at least 5-10 freshwater aquatic species, gathered from literature, QSAR, and ICE models, are compiled. A statistical distribution (e.g., log-normal) is fitted to this dataset [60].
- PNEC Calculation: The PNEC is derived from the SSD as the hazardous concentration for 5% of the species (HC~5~). This value is often divided by an assessment factor to ensure a sufficient margin of safety, resulting in the final PNEC value [60].
Risk Quotient (RQ) Calculation
The ecological risk is characterized using the Risk Quotient (RQ) method [60]: RQ = MEC / PNEC Where MEC is the Measured Environmental Concentration. An RQ < 0.1 indicates a low ecological risk, while an RQ ≥ 1.0 suggests a high potential risk that requires further attention [60].
Results and Data Analysis
Physicochemical Properties and Model Inputs
The following table summarizes the key properties of BPA and its alternatives, which are fundamental for QSAR predictions and understanding environmental fate.
Table 1: Physicochemical Properties of BPA and Its Alternatives [60]
| Chemical | Abbreviation | Molecular Weight (g/mol) | Water Solubility (mg/L) | Log Kow | Log Koc | Half-Life in Water (days) |
|---|---|---|---|---|---|---|
| Bisphenol A | BPA | 228.29 | 300 | 3.41 | 4.88 | 37.5 |
| Bisphenol S | BPS | 250.27 | 1100 | 1.65 | 2.5 | 37.5 |
| Bisphenol F | BPF | 200.24 | 540 | 2.91 | 4.47 | 15 |
Derived PNECs and Risk Characterization
The coupled in silico models enabled the derivation of PNECs and subsequent risk assessment for 32 major Chinese surface waters.
Table 2: Predicted No-Effect Concentrations (PNECs) and Risk Quotient (RQ) Ranges for BPA, BPS, and BPF in Surface Waters [60]
| Chemical | PNEC (μg/L) | RQ Range | Overall Ecological Risk Level |
|---|---|---|---|
| Bisphenol A (BPA) | 8.04 | ~0 to 1.86 | Low in most cases, but high in specific locations |
| Bisphenol S (BPS) | 35.2 | ~0 to 1.86 | Low in most cases, but high in specific locations |
| Bisphenol F (BPF) | 34.2 | ~0 to 1.86 | Low in most cases, but high in specific locations |
The data shows that BPA is the most potent of the three chemicals, with a PNEC approximately four times lower than those of BPS and BPF. While the overall ecological risk was low for most water bodies, the RQ values in specific locations like the Liuxi River, Taihu Lake, and Pearl River reached up to 1.86, indicating a high potential risk and demonstrating that the ecological risks posed by BPA alternatives have reached equivalent levels to those posed by BPA in some cases [60].
The Scientist's Toolkit: Essential Research Reagents and Platforms
The following table lists key computational tools and data resources essential for conducting in silico ecological risk assessments.
Table 3: Key Research Reagents and Computational Platforms for In Silico Risk Assessment
| Tool / Resource Name | Type | Function in Risk Assessment |
|---|---|---|
| VEGA Platform | QSAR Software | Provides multiple quantitative structure-activity relationship models to predict ecotoxicity endpoints based on chemical structure [60]. |
| USEPA Web-ICE | ICE Model Platform | Uses interspecies correlation estimation to predict toxicity for untested species from surrogate species data, expanding datasets for SSD [60]. |
| EPA CompTox Chemicals Dashboard | Chemical Database | Provides curated data on chemical structures, properties, and environmental fate, used as input for models [60]. |
| PubChem / ChemSpider | Chemical Databases | Public repositories for chemical information, including structures and physicochemical properties [60]. |
| ISPRI Toolkit | Immunoinformatics Platform | Used for assessing immunogenicity risk of biologics by identifying T-cell epitopes; an example of in silico tools for human health assessment [61]. |
Discussion on Regulatory Context and Pathways
The application of in silico models in regulatory decision-making is evolving. International guidelines increasingly advocate for non-animal testing methods, and models like QSAR and ICE are being applied in chemical risk assessment globally [60]. The case study's methodology aligns with initiatives like the European Partnership for the Assessment of Risks from Chemicals (PARC), which aims to close data gaps for BPA alternatives using advanced methods [62]. The relationship between the computational predictions and the broader regulatory risk assessment process is summarized below.
A key finding of the case study is the demonstration of regrettable substitution, where BPS and BPF, introduced as safer alternatives, were found to pose ecological risks comparable to BPA in certain environments [60]. This underscores the critical importance of thorough prospective risk assessments for replacement chemicals before they are widely adopted.
This case study successfully demonstrates that coupled in silico toxicology models (QSAR-ICE) provide an effective and reliable strategy for the ecological risk assessment of data-poor chemicals, specifically BPA alternatives. The models generated sufficient data to construct SSDs and derive PNECs, revealing that while the overall risk in Chinese surface waters is low, localized high risks exist for BPA, BPS, and BPF. This evidence justifies the need for increased attention to these emerging contaminants. The integrated workflow presented offers a time- and resource-efficient path forward for screening the ecological risk of new chemicals, supporting safer substitution principles and informed environmental management decisions.
The Adverse Outcome Pathway (AOP) framework is a conceptual construct that facilitates the organization and interpretation of mechanistic data across multiple biological levels, deriving from a range of methodological approaches including in silico, in vitro, and in vivo assays [17]. An AOP depicts a sequential chain of causally linked events at different levels of biological organization, beginning with a Molecular Initiating Event (MIE), where a chemical stressor interacts with a biological target, and progressing through a series of Key Events (KEs) that lead to an Adverse Outcome (AO) of regulatory relevance [63]. This framework serves as a knowledge assembly and translation tool, designed to support the use of pathway-specific mechanistic data for predicting risks posed by chemicals to human health and the environment [64]. The AOP framework is chemically agnostic, meaning it captures biological response-response relationships that can be initiated by any number of chemical or non-chemical stressors sharing a common MIE [63] [64].
In the context of in silico environmental risk assessment research, AOPs provide a biological scaffold that enables the translation of data from high-throughput screening assays and computational models into predictions of adverse effects relevant to regulatory decision-making [65] [64]. The quantification of AOPs, leading to the development of quantitative AOPs (qAOPs), is a critical step towards more reliable prediction of chemically induced adverse effects [17]. QAOPs are toxicodynamic models that formalize the quantitative relationships between KEs, allowing for the prediction of the likelihood and severity of the AO based on the intensity of perturbations at earlier, more readily measurable events in the pathway [17]. This quantitative understanding is essential for moving from qualitative hazard identification to quantitative risk assessment, thereby linking mechanistic data directly to decisions in chemical safety assessment [17].
The Conceptual Integration of In Silico Models and AOPs
The integration of in silico models and the AOP framework creates a powerful synergy for identifying and evaluating novel biomarkers. In silico toxicology integrates different mathematical and computational models to predict chemical toxicity based on patterns of structural and physicochemical properties related to toxicological activity [65]. When combined with the AOP framework, these models gain a structured biological context, enhancing their mechanistic interpretability and predictive capacity for hazard assessments [65]. This integration is pivotal for addressing the challenge of assessing the vast number of chemicals with limited or no hazard information, as it allows for the efficient prioritization of substances for further testing [64].
The AOP framework's modular structure, composed of MIES, KEs, and KERs, provides an ideal platform for organizing the complex data generated by in silico approaches. A KE is a measurable biological change that is essential to the progression of a defined biological perturbation [63]. As such, KEs represent candidate biomarkers—measurable indicators of biological processes, pathogenic states, or pharmacological responses to therapeutic interventions [65]. The Key Event Relationships (KERs) describe the causal linkages between these biomarkers, establishing a chain of evidence connecting a molecular-level perturbation to an adverse outcome at the organism or population level [63]. By exploring AOP networks, it becomes possible to identify measurable KEs to be used as biomarkers of exposure or effect and to understand their interrelationships through KERs [65]. This structured approach supports the use of a Weight of Evidence (WoE) approach for judging the confidence in each proposed relationship based on biological plausibility, empirical support, and essentiality [65].
Table 1: Core Components of the AOP Framework and Their Relationship to Biomarkers
| AOP Component | Definition | Role in Biomarker Identification |
|---|---|---|
| Molecular Initiating Event (MIE) | The initial interaction between a stressor and a biological target within an organism [63]. | Identifies the most proximal biomarker of exposure and potential target for in silico prediction. |
| Key Event (KE) | A measurable biological change at different levels of organization essential to the progression towards the AO [63]. | Represents a candidate biomarker of effect; can be measured by in chemico, in vitro, or in vivo assays. |
| Adverse Outcome (AO) | A change in organisms or populations considered relevant for regulatory decision-making [63]. | Defines the distal phenotypic anchor for biomarker validation. |
| Key Event Relationship (KER) | A scientifically-based, causal linkage between two KEs [63]. | Informs the predictive relationship between biomarkers, enabling inference of distal events from proximal measurements. |
Methodological Workflow: From AOP Development to Biomarker Discovery
The process of linking AOPs with in silico predictions to identify biomarkers follows a structured workflow. This workflow integrates computational and experimental methods to build confidence in the proposed biomarkers and their place within a toxicological pathway.
AOP Development and Network Analysis
The initial phase involves the identification and assembly of existing AOPs related to the toxicological endpoint of interest from knowledge bases such as the AOP-Wiki [65]. For complex endpoints, multiple AOPs can be assembled into an AOP network that captures shared KEs and KERs, providing a more comprehensive view of the biological system [63]. As demonstrated by Spinu et al. (2019), generating and analyzing an AOP network allows for the identification of the most common and highly connected KEs, which represent strong candidates for biomarker development due to their central role in the toxicological response [65]. This network analysis helps prioritize which KEs to test and provides a basis for subsequent quantitative modeling.
In Silico Model Application for MIE and KE Prediction
With a structured AOP as a scaffold, in silico models are deployed to predict the MIE and subsequent KEs. Alert-based models, such as quantitative structure-activity relationship (QSAR) models, can predict whether a chemical has structural features associated with a specific MIE, like covalent protein binding or receptor activation [65]. More advanced machine learning (ML) models can be trained on high-throughput screening data or 'omics datasets to predict intermediate KEs. For instance, ensemble ML platforms can integrate multiple predictor variables, such as intracellular protein expression and blood cell counts, to classify exposure status or quantitatively estimate received dose, as seen in radiation biodosimetry studies [66]. The use of feature selection algorithms, like Boruta, within these ML workflows helps identify the most influential predictor variables, thereby refining the panel of candidate biomarkers [66].
Quantitative AOP (qAOP) Modeling and Biomarker Validation
The qualitative AOP is then transformed into a qAOP by defining the quantitative relationships between KEs [17]. This involves gathering existing data or generating new experimental data to model the dynamics of the pathway. The essentiality of a proposed biomarker (KE) is tested by determining if blocking or modulating that event prevents the downstream KEs and the AO [65]. This can be achieved through targeted in vitro or in vivo experiments using genetic knockdown, pharmacological inhibitors, or other interventional strategies. A successful example is the qAOP for skin sensitization, which incorporates several intermediate KEs related to inflammatory cytokines and T-cell proliferation. This AOP forms the basis for a validated suite of in vitro assays that can replace traditional in vivo tests for predicting sensitization potential [64].
The following diagram illustrates the core logical structure of an AOP and its key components:
Diagram 1: Core AOP structure showing MIE, KEs, KERs, and AO.
Case Studies and Experimental Data
Case Study 1: Radiation Biodosimetry via a Biomarker Panel and Machine Learning
A compelling example of an integrated approach is the validation of a blood biomarker panel for radiation biodosimetry. The study aimed to develop a high-throughput bioassay for rapid and individualized radiation dose assessment up to 7 days post-exposure [66]. The candidate biomarkers included intracellular leukocyte proteins (ACTN1, DDB2, FDXR) known to play roles in DNA Damage Response (DDR) mechanisms, along with blood cell counts (CD19+ B-cells and CD3+ T-cells) [66].
Experimental Protocol: Juvenile and adult C57BL/6 mice were total-body irradiated with 0, 1, 2, 3, or 4 Gy. Peripheral blood was collected 1, 4, and 7 days post-exposure. Biomarkers were quantified using imaging flow cytometry (IFC): surface-stained B- and T-cells for cell counts, and intracellular staining for protein expression levels measured by Mean Fluorescence Intensity (MFI) [66].
Data Integration and Modeling: An ensemble machine learning platform was employed. The Boruta feature selection algorithm first identified the most influential predictor variables. A stacking ensemble then aggregated the predictive power of these variables using multiple ML methods to generate two outputs: 1) classification of exposure status, and 2) quantitative dose reconstruction [66].
Results and Relevance to AOPs: This approach successfully classified exposure (ROC AUC = 0.94) and reconstructed dose (R² = 0.79, RMSE = 0.68 Gy) [66]. The identified biomarkers (ACTN1, DDB2, FDXR, lymphocyte depletion) can be viewed as Key Events within an AOP for radiation-induced hematopoietic syndrome. The ML model effectively functions as a quantitative, multi-parameter KER, integrating measurements from several KEs to predict the probability and severity of the overarching adverse outcome.
Table 2: Key Biomarkers and Performance Metrics from Radiation Biodosimetry Case Study
| Biomarker | Biological Function / Role | Measurement Technique | Key Finding |
|---|---|---|---|
| ACTN1 | Cross-linking cytoskeletal protein; associated with stress-induced cellular senescence [66]. | Imaging Flow Cytometry (MFI) | Part of a panel enabling dose reconstruction up to 7 days post-exposure. |
| DDB2 | DNA lesion recognition protein for nucleotide excision repair; mediates apoptosis/senescence [66]. | Imaging Flow Cytometry (MFI) | Part of a panel enabling dose reconstruction up to 7 days post-exposure. |
| FDXR | Mitochondrial flavoprotein; modulates p53-dependent apoptosis [66]. | Imaging Flow Cytometry (MFI) | Part of a panel enabling dose reconstruction up to 7 days post-exposure. |
| CD19+ B-cells & CD3+ T-cells | Mature lymphocytes, highly radiosensitive cell types [66]. | Imaging Flow Cytometry (% of cells) | Part of a panel enabling dose reconstruction up to 7 days post-exposure. |
| Ensemble ML Model | Integrates all biomarker data for prediction. | Stacking algorithm with feature selection | Performance: Exposure Classification: ROC AUC = 0.94. Dose Reconstruction: R² = 0.79, RMSE = 0.68 Gy [66]. |
Case Study 2: Predictive Models for Metastatic Colorectal Cancer (mCRC) Therapy
In a clinical context, a research protocol aims to use AI to predict therapy response in mCRC patients. The unmet need is the lack of clinical tools to identify responders to specific regimens before treatment initiation [67]. The study uses a molecular biomarker signature as inputs for ML models.
Experimental Protocol: The study involves a retrospective analysis of formalin-fixed, paraffin-embedded (FFPE) tumor samples from mCRC patients. Molecular biomarkers examined include chromosomal instability, mutational profiles of 50 CRC-related genes, and whole transcriptome expression (including long non-coding RNAs) [67].
Data Integration and Modeling: Data from public archives (TCGA, GEO) and a retrospective cohort are synthesized. Machine learning technology (e.g., random survival forest, neural networks) is used to develop and validate a predictive model that classifies patients as responders or non-responders to chemotherapy, alone or with targeted therapy [67]. The model's performance is evaluated using sensitivity, specificity, and AUC [67].
Results and Relevance to AOPs: While not presented as a traditional AOP, the underlying principle is congruent. The molecular features (e.g., specific mutations, chromosomal instability, lncRNA signatures) can be viewed as MIEs or early KEs in AOP networks leading to the adverse outcomes of treatment failure or disease progression. The ML model identifies a predictive signature—a set of interconnected biomarkers—whose relationship to the clinical outcome is learned from the data, effectively embodying a complex, quantitative KER.
The workflow for integrating diverse data types to build such predictive models is shown below:
Diagram 2: ML workflow for predicting therapy response from biomarker data.
The Scientist's Toolkit: Essential Reagents and Methods
Implementing the integrated AOP/in silico approach requires a specific toolkit of research reagents and methodologies. The table below details key materials and their functions based on the cited research.
Table 3: Essential Research Reagents and Methods for AOP-In Silico Biomarker Studies
| Tool / Reagent | Function / Application | Example from Literature |
|---|---|---|
| Imaging Flow Cytometry (IFC) | High-throughput quantification of both surface markers (cell counts) and intracellular protein biomarkers in single cells [66]. | Used to measure ACTN1, DDB2, FDXR MFI and % B-/T-cells in mouse blood for radiation biodosimetry [66]. |
| High-Throughput In Vitro Assays | Rapid screening of chemical bioactivity and cytotoxicity in cell lines. | Miniaturized OECD TG 249 assay in RTgill-W1 cells for fish ecotoxicology; Cell Painting assay for phenotypic screening [9]. |
| Next-Generation Sequencing (NGS) | Comprehensive analysis of molecular biomarkers including mutational profiles and transcriptome-wide expression. | Analysis of mutational status of 50 genes and whole-transcriptome via Affymetrix arrays in mCRC FFPE samples [67]. |
| Formalin-Fixed Paraffin-Embedded (FFPE) Samples | Archival source of clinical tissue for retrospective analysis of molecular biomarkers. | Used to extract DNA/RNA for profiling chromosomal instability, mutations, and lncRNA in mCRC patients [67]. |
| Machine Learning Algorithms | Developing predictive models by integrating multiple biomarker inputs; includes feature selection and ensemble methods. | Boruta algorithm for feature selection and stacking ensemble for dose prediction in radiation study [66]; XGBoost for CRC risk stratification [68]. |
| In Vitro Disposition (IVD) Model | An in silico model that predicts freely dissolved concentrations of chemicals in vitro, accounting for sorption to plastic and cells [9]. | Improved concordance between in vitro bioactivity (PACs) and in vivo fish toxicity data [9]. |
The integration of the AOP framework with in silico predictions represents a paradigm shift in toxicological research and biomarker discovery. This synergistic approach provides a structured, mechanistic basis for interpreting complex data from new approach methodologies (NAMs), thereby enhancing the reliability and regulatory acceptance of predictive toxicology [17] [64]. By viewing biomarkers as measurable Key Events within causal pathways, researchers can move beyond simple correlative associations to establish a functional understanding of their role in disease etiology or toxicological processes. The future of this field lies in the continued development of quantitative AOP networks, the refinement of machine learning models for pathway-based prediction, and the collaborative building of a centralized knowledge base to support more efficient, evidence-based environmental risk assessment and personalized medicine [17] [65] [69].
Navigating Challenges and Enhancing Model Robustness for Regulatory Acceptance
In silico methods, encompassing computational approaches like quantitative structure-activity relationships (QSAR), machine learning (ML) models, and toxicokinetic-toxicodynamic (TK-TD) modeling, are reshaping the landscape of environmental risk assessment (ERA) for chemicals [57] [70]. These New Approach Methodologies (NAMs) offer powerful tools for predicting the environmental fate and ecological effects of substances, from pesticides to industrial chemicals, with the goals of improving efficiency, reducing animal testing, and handling complex data [70] [2]. The integration of artificial intelligence, particularly machine learning, has enabled the analysis of complex, high-dimensional datasets that characterize modern chemical and toxicological research [57]. However, the translation of these computational predictions into reliable regulatory decisions and scientific insights is hampered by three interconnected core limitations: applicability domain, data biases, and mechanistic uncertainties. A bibliometric analysis of the field reveals an exponential publication surge from 2015, dominated by environmental science journals, with China and the United States leading research output, indicating the field's rapid growth and global importance [57]. Effectively addressing these limitations is crucial for strengthening the regulatory acceptance and scientific robustness of in silico environmental risk assessment.
The Applicability Domain Limitation
Definition and Significance
The Applicability Domain (AD) represents the response and chemical structure space for which a given in silico model generates reliable predictions [71]. It is fundamentally based on the principle that a model is inherently constrained by the information within its training set; a model developed on specific substance classes may perform poorly when applied to chemicals outside that structural domain [71]. Regulatory authorities, including those implementing the European REACH regulation, require the evaluation of a model's applicability domain, making it a critical component for regulatory acceptance [71]. The AD essentially defines the boundaries within which the model can be trusted, and assessing whether a target substance falls within these boundaries is paramount for generating reliable predictions for environmental risk assessment.
Underlying Causes and Challenges
The challenge of defining the AD stems from the chemical diversity of substances potentially subjected to prediction. Modern models are often built on heterogeneous training sets, but these sets can never encompass all possible chemical variations [71]. Some chemical families exhibit peculiar behaviors that are poorly represented in training data, meaning the model cannot adequately learn to predict their effects. Furthermore, different software platforms use varied approaches to measure AD, leading to inconsistencies; some provide a simple binary outcome (inside/outside AD), while others, like the VEGA platform, employ a more nuanced, quantitative Applicability Domain Index (ADI) [71]. This lack of standardization complicates the comparison of predictions across different tools and platforms.
Quantitative Assessment of Model Performance and AD
The VEGA tool's quantitative approach to AD demonstrates its impact on prediction confidence. The platform evaluates chemical structures and other endpoint-specific features to measure the applicability domain, enabling users to identify less accurate predictions.
Table 1: Impact of VEGA's Applicability Domain Index (ADI) on Model Performance
| Model Type | Endpoint Category | Performance on Full Test Set (Accuracy) | Performance on ADI-Filtered Set (Accuracy) | Key AD Assessment Features |
|---|---|---|---|---|
| Classification Models | Human Toxicity | Variable across models | Significant improvement | Chemical similarity, presence of toxicity alerts |
| Classification Models | Ecotoxicity | Variable across models | Significant improvement | Structural features, mechanistic alerts |
| Classification Models | Environmental Fate | Variable across models | Significant improvement | Property-specific chemical descriptors |
| Regression Models | Toxicokinetic Properties | Lower correlation coefficients | Higher correlation coefficients | Similarity-based prediction accuracy |
The data shows that filtering predictions based on the ADI tool successfully identifies and removes less reliable predictions, thereby increasing the overall accuracy and confidence in the results for the remaining compounds [71]. This process involves checking the chemical similarity between the target substance and the training set compounds, and comparing predictions for similar substances to identify potential inconsistencies [71].
Experimental Protocol for AD Evaluation
Protocol: Characterizing the Applicability Domain of a QSAR Model
- Define the Chemical Space: Using the model's training set, calculate the range and distribution of chemical descriptors (e.g., molecular weight, log P, topological surface area) to establish the model's fundamental chemical domain.
- Assess Structural Similarity: For the target compound, calculate its similarity to the training set compounds using appropriate metrics (e.g., Tanimoto coefficient based on fingerprints). Tools like the VEGA platform automate this process and provide a quantitative measure [71].
- Check for Extrapolation: Determine if the target compound's descriptor values lie within the multivariate space defined by the training set. This can involve leverage-based approaches or distance-to-model metrics.
- Evaluate Mechanistic Consistency: Verify if the target compound contains structural features or alerts associated with the predicted endpoint that are consistent with the model's underlying mechanics.
- Apply a Quantitative AD Index: Utilize a tool like VEGA's ADI, which consolidates multiple checks (similarity, reliability of similar compounds' data, consistency of predictions) into a single, interpretable value to guide the user on prediction reliability [71].
Data Biases Limitation
Typology of Data Biases
In silico toxicology methods are susceptible to various data biases that can systematically distort predictions and compromise their validity for risk assessment. These biases can be categorized based on their origin in the data lifecycle. Selection bias arises from systematic differences in how data is selected or generated, such as non-random allocation of test systems or over-representation of certain chemical classes in training data [72]. Reporting bias occurs when the reporting of scientific results is influenced by the nature and direction of the findings, leading to incomplete or selective data availability for model training [72]. Furthermore, algorithmic bias can be introduced by the machine learning models themselves, often reflecting and amplifying imbalances present in the training data [72]. A bibliometric analysis of ML in environmental chemical research revealed a significant 4:1 bias in keyword frequencies toward environmental endpoints over human health endpoints, highlighting a domain-specific data imbalance in the field [57].
Impact on Model Reliability and Risk Assessment
Data biases directly threaten the internal validity of the studies used to build predictive models, affecting the cause-and-effect relationship between a chemical and its toxicological outcome [72]. In a regulatory context, models trained on biased data can lead to an overestimation or underestimation of the true environmental risk, resulting in flawed regulatory decisions and inadequate environmental protection [73] [72]. For instance, if a model for predicting pesticide toxicity to bees is trained predominantly on data from a single chemical class (e.g., organophosphates), its predictions for a newer class of insecticides (e.g., neonicotinoids) may be unreliable, failing to identify a significant ecological threat [2]. The issue is compounded by the matrix influence, trace concentration, and complex scenarios that are often ignored in laboratory-based training data, limiting the model's applicability to real-world environmental conditions [73].
Framework for Uncertainty Categorization
A systematic framework is needed to identify and categorize sources of uncertainty, including those stemming from data biases, to improve the transparency and evaluation of in silico methods.
Experimental Protocol for Bias Assessment
Protocol: Assessing Risk of Bias in Data for In Silico Models
- Problem Formulation: Define the intended use of the model and the regulatory context, identifying which types of bias are most critical for the endpoint and chemical space of interest [72] [74].
- Data Provenance Analysis: Trace the origin of the data in the training set. Use specialized tools like the SYRCLE (for in vivo studies) or OHAT frameworks to assess the risk of bias in the underlying toxicological studies [72].
- Chemical Space Representation Analysis: Evaluate the diversity of the training set. Identify over- and under-represented chemical classes, and assess whether the target chemical for prediction falls into a data-poor region [73].
- Sensitivity Analysis: Test the model's performance on held-out test sets that are stratified to represent different chemical classes or experimental conditions. Significant performance variations indicate potential data biases [74].
- Application of AI-Based Bias Detection: Leverage emerging AI tools designed to screen studies, detect anomalies, and identify patterns indicative of selection, performance, or reporting biases in large datasets [72].
Mechanistic Uncertainties Limitation
Mechanistic uncertainties arise from an incomplete understanding of the biological and toxicological processes that link chemical exposure to an adverse ecological outcome. While in silico models, particularly machine learning, excel at identifying correlations between chemical structures and toxicological endpoints, they often function as "black boxes" with limited mechanistic interpretability [75] [73]. This lack of transparency makes it difficult to understand why a particular prediction was made, which erodes confidence, especially for novel chemicals or unexpected results. A key challenge is the complexity of transformation products (TPs); chemicals can undergo (a)biotic degradation in the environment, forming TPs whose identity, toxicity, and behavior are poorly characterized and difficult to predict [75]. Mechanistic uncertainties are also inherent in extrapolating across species and biological levels of organization, for example, when predicting population-level effects from individual-level toxicity data [70].
Consequences for Predictive Toxicology
The primary consequence of mechanistic uncertainty is limited regulatory acceptance and hesitancy to base significant risk management decisions solely on in silico predictions [75] [46]. Without a plausible biological mechanism, it is challenging to build the weight-of-evidence necessary for robust risk assessment. Furthermore, models with high mechanistic uncertainty are prone to failure when applied to chemicals that act through novel or unpredicted pathways of toxicity. This is particularly problematic for emerging contaminants like microplastics and per-/polyfluoroalkyl substances (PFAS), where the underlying toxicological mechanisms are still an active area of research [57] [73]. Models may also struggle to accurately predict effects from chemical mixtures, where interactions between compounds can lead to non-additive effects that are difficult to anticipate without a deep mechanistic understanding [70].
Confidence Framework for Predictions
To manage mechanistic uncertainty, a framework of confidence levels for in silico predictions has been proposed, helping to communicate reliability and guide use [75].
Table 2: Confidence Levels for In Silico Transformation Product Prediction and Toxicological Assessment
| Confidence Level | Mechanistic Understanding | Data Support | Recommended Use |
|---|---|---|---|
| High | Well-established Adverse Outcome Pathway (AOP); known molecular initiating event. | Experimental data for parent compound and analogous TPs; robust QSAR model within its AD. | Can support regulatory prioritization and decision-making. |
| Medium | Plausible mechanism based on structural alerts or read-across; limited AOP support. | Limited experimental data for TPs; consensus predictions from multiple in silico tools. | Useful for screening and hypothesis generation; requires further evidence. |
| Low | Putative mechanism only; unknown if the predicted TP forms in relevant environments. | No experimental data for TPs; reliance on a single rule-based model. | For internal prioritization only; not sufficient for regulatory purposes. |
| Very Low | No plausible mechanism; prediction is purely statistical. | Model used outside its applicability domain; conflicting predictions. | Not recommended for any application; requires generation of new data. |
Experimental Protocol for Mechanistic Evaluation
Protocol: Integrating Mechanistic Understanding into In Silico Predictions
- Adverse Outcome Pathway (AOP) Development: Frame the prediction within the context of an existing AOP. If an AOP is available, it provides a structured, mechanistic foundation linking a molecular initiating event to an adverse outcome at the individual or population level [70] [46].
- Structural Alert Analysis: Use software like the OECD QSAR Toolbox to identify known structural features associated with toxicity (structural alerts) in the target compound. This links the prediction to established mechanistic knowledge [70].
- In Vitro-In Vivo Extrapolation (IVIVE): For higher-tier assessments, integrate in silico predictions with data from in vitro bioassays using quantitative in vitro to in vivo extrapolation (QIVIVE) models. This helps bridge the gap between high-throughput screening data and relevant ecological doses [70].
- Toxicokinetic-Toxicodynamic (TK-TD) Modeling: Employ biologically-based models, such as Dynamic Energy Budget (DEB) models, to connect external exposure to internal dose (TK) and then to toxicological effects over time (TD). This provides a more mechanistically realistic prediction of toxicity than simple endpoint-prediction models [70].
- Consensus Modeling and Weight-of-Evidence: Generate predictions using multiple, independent in silico methods (e.g., a QSAR model, a rule-based system, and a read-across approach). A consensus among different methods increases confidence in the prediction and helps mitigate the uncertainty of any single approach [75] [46].
Successfully navigating the limitations of in silico ERA requires a suite of software, databases, and conceptual tools.
Table 3: Key Resources for Addressing In Silico Limitations
| Tool/Resource Name | Type | Primary Function | Role in Mitigating Limitations |
|---|---|---|---|
| VEGAHUB | Software Platform | Provides access to >100 (Q)SAR models for human health, ecotoxicity, and environmental fate endpoints. | Its quantitative Applicability Domain Index (ADI) directly addresses the Applicability Domain limitation [71]. |
| OECD QSAR Toolbox | Software Platform | Supports chemical grouping, read-across, and (Q)SAR model development, integrating multiple databases and tools. | Helps identify profilers and structural alerts, reducing mechanistic uncertainty and data gaps via read-across [70]. |
| US-EPA CompTox Chemicals Dashboard | Database | A large database (~1.2M substances) with physicochemical, fate, hazard, exposure, and in vitro bioactivity data. | Provides a vast data source for model training and testing, helping to identify and rectify data biases [70]. |
| Adverse Outcome Pathway (AOP) Wiki | Knowledge Base | A collaborative repository of AOPs that describe mechanistic linkages across biological levels of organization. | Provides a structured framework for building mechanistic understanding, reducing mechanistic uncertainties [46]. |
| SYRCLE/OHAT Tools | Risk of Bias Tool | Specialized frameworks for assessing the risk of bias in in vivo (SYRCLE) and human (OHAT) studies. | Allows for the critical appraisal of underlying data quality, directly addressing data biases [72]. |
| TK-PBPK Modeling Platforms | Modeling Framework | Platforms for developing Toxicokinetic (TK) and Physiologically-Based Pharmacokinetic (PBPK) models. | Enables extrapolation from external dose to internal target site concentration, refining dose-response and reducing uncertainty [70] [46]. |
Integrated Workflow for a Robust In Silico Assessment
To effectively manage the triad of limitations in practice, researchers should adopt an integrated workflow that incorporates checks and balances at each stage. The following diagram outlines a robust protocol for generating and evaluating in silico predictions, from problem formulation to final reporting.
This workflow emphasizes that in silico assessment is an iterative process. If a prediction is found to be outside the model's Applicability Domain, is based on biased data, or lacks mechanistic plausibility, the process should loop back to an earlier stage, potentially requiring the selection of a different model or the generation of new data. The final step of reporting with a clear statement of uncertainty is essential for transparent and scientifically honest communication.
In the field of environmental risk assessment, a significant challenge is the pervasive lack of complete experimental toxicological data for a vast number of chemicals in commerce. For the leather and textile industry (LTI) alone, it is estimated that over 10,000 substances are used, many of which lack comprehensive hazard characterization [76]. This data gap presents a major obstacle for regulators and industries committed to protecting human and environmental health, particularly under stringent regulatory frameworks like the European Union's Registration, Evaluation, Authorisation and restriction of CHemicals (REACH) and the Chemicals Strategy for Sustainability. A complete experimental evaluation of all these substances would require an enormous investment of time and money, and would necessitate an ethically unacceptable number of in vivo experiments [76]. This reality has accelerated the development and adoption of New Approach Methodologies (NAMs), particularly in silico methods, which offer a pathway to characterize relevant toxicological endpoints with less time and financial investment while aligning with the 3Rs (Replacement, Reduction, and Refinement) principle for animal testing [76] [28].
The data gap problem extends beyond merely identifying hazardous substances. Effectively assessing chemicals also requires understanding their environmental fate, including the formation and behavior of transformation products (TPs). These TPs form through (a)biotic processes and can be detected in environmental concentrations comparable to or even exceeding their parent compounds, indicating significant toxicological relevance [75]. However, identifying them is challenging due to the complexity of transformation processes and insufficient data, creating a secondary layer of data gaps that complicate comprehensive risk assessment [75]. This whitepaper outlines a structured, multi-faceted strategy to address these data gaps through integrated in silico methodologies, providing researchers with a robust framework for chemical safety assessment.
Foundational In Silico Methodologies
In silico toxicology encompasses a suite of computational methods used to predict the properties, hazard, and risk of chemicals. These methodologies can be broadly categorized into the following approaches.
(Quantitative) Structure-Activity Relationships ((Q)SAR)
(Q)SAR models establish mathematical relationships between the chemical structure of a compound (descriptors) and its biological activity or property [77]. These models can be used for a range of predictions, from categorical classification (e.g., persistent vs. not persistent) to continuous values (e.g., binding affinity) [78].
- Application: REACH legislation explicitly encourages the use of (Q)SAR to fill data gaps for toxicity, fate, and physicochemical properties, especially when experimental data is unavailable [77].
- Development: A classic workflow involves compiling a dataset with experimental half-life values for environmental compartments like sediment, soil, and water. This dataset is split into training and test sets, after which a machine learning algorithm, such as k-Nearest Neighbor (k-NN), is applied to build a predictive model [77].
Read-Across
Read-across is a data-gap filling technique that involves using data from one or more similar source substances (the analogues) to predict the same property for a target substance that lacks data. The underlying principle is that similar chemicals are likely to have similar properties and biological activities.
- Application: It is particularly valuable for evaluating groups of chemicals, such as those within a specific chemical class, and for endpoints where robust (Q)SAR models are not available [76].
- Workflow: The process involves identifying a group of structurally similar chemicals for which experimental data exists. The property of interest for the data-poor target chemical is then inferred based on the data from these analogues.
Molecular Docking and Dynamics
These are structure-based methods that simulate the interaction between a chemical and a biological macromolecule, such as a protein receptor.
- Molecular Docking predicts the preferred orientation of a small molecule (ligand) when bound to its target, which is used to predict the binding affinity and mode of action [78] [79].
- Molecular Dynamics (MD) simulations take this further by simulating the physical movements of atoms and molecules over time, providing insights into the stability and kinetics of the binding interaction under more realistic conditions [79].
Table 1: Core In Silico Methodologies for Filling Data Gaps
| Methodology | Underlying Principle | Primary Application | Key Advantage |
|---|---|---|---|
| (Q)SAR [77] | Correlates molecular descriptors with biological activity. | Predicting continuous or categorical toxicological endpoints (e.g., carcinogenicity, persistence). | High-throughput screening of large chemical libraries. |
| Read-Across [76] | Infers properties based on data from structurally similar compounds. | Filling data gaps for endpoints without robust (Q)SAR models. | Does not require a pre-existing model; conceptually simple. |
| Molecular Docking [78] [79] | Predicts ligand binding orientation and affinity to a protein target. | Identifying potential molecular initiating events and off-target effects. | Provides mechanistic insight into bioactivity. |
| Machine Learning [76] [78] | Algorithms autonomously learn complex patterns from large, diverse datasets. | Integrating multimodal data (chemical, genomic, phenotypic) for DTI and hazard prediction. | Capable of modeling non-linear relationships and handling high-dimensional data. |
An Integrated Strategy for Assessment and Prioritization
To tackle the data gap problem systematically, isolated in silico methods must be integrated into a cohesive assessment and prioritization strategy. The following workflow outlines a proven, step-by-step process.
Compilation and Annotation of a Chemical Inventory
The first step is to create a comprehensive catalogue of substances of interest. In a project for the leather and textile industry, researchers started with an initial set of 3,464 substances and expanded it by integrating data from multiple sources [76]. Unique identifiers like CAS Registry Numbers (CAS-RN) are crucial for tracking substances across databases. Each substance in the inventory is then annotated with any available experimental toxicological information from public sources such as ECHA (REACH, CLP), Pharos, and PubChem [76]. This process clarifies the knowns and unknowns.
In Silico Prediction and Data Gap Filling
For endpoints where experimental data is missing, in silico models are deployed. In the LTI case study, this approach allowed for the prediction of approximately 6,483 properties that were not available from existing data sources [76]. The endpoints of high regulatory concern, such as CMR (Carcinogenic, Mutagenic, Reprotoxic) properties, endocrine disruption (ED), and PBT/vPvB (Persistent, Bioaccumulative and Toxic/very Persistent and very Bioaccumulative) are primary targets for such predictions [76] [77]. For PBT assessment, an integrated strategy can be employed, where separate models for persistence in sediment, soil, and water are built and then combined into a final, conservative overall assessment [77].
Integrated Assessment and Confidence Scoring
Predictions from various models and for different endpoints must be synthesized to reach an overall conclusion. This can be achieved through decision workflows that integrate the results [76]. For example, a decision tree might combine predictions for sediment, water, and soil persistence with identified structural alerts to yield a final classification [77]. Given the uncertainties inherent in any model, it is critical to assign a level of confidence to the predictions. A proposed framework involves using four distinct confidence levels, which help communicate the reliability of the in silico assessment and guide its use in decision-making [75]. Expert judgment remains essential for interpreting the integrated results.
Experimental Validation and Confidence Building
In silico predictions require validation to build scientific and regulatory confidence. A prospective validation exercise, where computational predictions are compared against newly generated experimental data, is a robust approach [76].
Protocol for Prospective Validation
- Selection of Compounds: Choose a representative subset of chemicals from the inventory. These should be selected to represent different reliability scenarios, covering various chemical classes and predicted outcome categories (e.g., predicted positive, predicted negative, borderline) [76].
- Experimental Testing: Subject the selected compounds to standardized, OECD-approved experimental assays for the endpoints of interest [76]. For toxicological endpoints, this may include in vitro assays for cytotoxicity, genotoxicity, or endocrine disruption potential. For environmental fate, ready biodegradability tests can serve as a screening tool for persistence [77].
- Comparison and Model Refinement: Compare the experimental results with the in silico predictions. Metrics such as accuracy, sensitivity, specificity, and Matthews Correlation Coefficient (MCC) can be used for categorical models [77]. Discrepancies between prediction and experiment should be analyzed to identify limitations in the models and guide their refinement in an iterative process of continuous learning [76].
Table 2: Example Performance Metrics from a k-NN Model for Persistence
| Environmental Compartment | Training Set Accuracy | Test Set Accuracy | Model Type | Application |
|---|---|---|---|---|
| Sediment [77] | > 0.79 | > 0.76 | k-Nearest Neighbor (k-NN) | Classifying persistence under REACH |
| Soil [77] | > 0.79 | > 0.76 | k-Nearest Neighbor (k-NN) | Classifying persistence under REACH |
| Water [77] | > 0.79 | > 0.76 | k-Nearest Neighbor (k-NN) | Classifying persistence under REACH |
Implementing a successful in silico strategy requires a combination of data resources, software tools, and computational frameworks. The table below details key reagents and tools for the modern computational toxicologist.
Table 3: Essential Toolkit for In Silico Chemical Risk Assessment
| Tool Category | Example Tools & Databases | Function and Application |
|---|---|---|
| Chemical Databases [76] | ECHA, Pharos, PubChem | Sources for chemical structures, identifiers, and experimental hazard annotations. |
| Toxicology Databases [76] | EPA Genetox, REACH Dossiers | Provide curated experimental data for model training and validation. |
| QSAR/Modeling Software [77] [80] | SARpy, KNIME, Orange Data Mining | Used to identify structural alerts, build predictive models, and create analysis workflows. |
| Structure-Based Design [79] [80] | AutoDock Vina, GROMACS, PyMol | Performs molecular docking, dynamics simulations, and protein structure visualization. |
| Programming Libraries [76] [78] | RDKit, Scikit-learn, Deep Learning Frameworks (PyTorch, TensorFlow) | Provide cheminformatics functionality and machine learning algorithms for custom model development. |
The data gap problem for chemicals is too vast to be solved by traditional experimental approaches alone. The strategic integration of in silico methodologies offers a viable, efficient, and ethically preferable path forward. As demonstrated in various industrial and regulatory contexts, a continuous learning strategy that combines computational predictions with targeted experimental validation can effectively characterize chemical hazards, prioritize substances for further action, and support the transition to safer and more sustainable chemicals [76] [28]. The future of environmental risk assessment lies in the continued refinement of these computational pipelines, the expansion of high-quality data for model training, and the development of standardized frameworks for evaluating and communicating prediction confidence, ultimately enabling a more proactive and preventative approach to chemical safety.
In silico environmental risk assessment (ERA) represents a paradigm shift in ecotoxicology, leveraging computational models to predict the harmful effects of chemicals on the environment. This approach has gained significant traction as regulatory agencies and researchers seek to overcome the limitations of traditional animal testing while addressing the vast number of chemicals requiring assessment. The coupling of Quantitative Structure-Activity Relationship (QSAR) and Interspecies Correlation Estimation (ICE) models has emerged as a particularly powerful methodology for generating comprehensive toxicity datasets where experimental data are limited or absent. These coupled models enable researchers to conduct robust risk assessments for emerging contaminants while adhering to the principles of replacement, reduction, and refinement of animal testing (the 3Rs) [1] [13].
The fundamental premise of model coupling lies in the complementary strengths of QSAR and ICE approaches. QSAR models predict chemical toxicity based on molecular structure and physicochemical properties, while ICE models extrapolate known toxicity values from tested species to untested species. When integrated, these approaches facilitate the development of Species Sensitivity Distributions (SSDs), which are essential for deriving Predicted No-Effect Concentrations (PNECs) and establishing Water Quality Criteria (WQC) [1] [81]. This technical guide explores the principles, methodologies, and applications of coupled QSAR-ICE models, providing researchers with a comprehensive framework for implementing this approach in environmental risk assessment.
Theoretical Foundations of QSAR and ICE Models
Quantitative Structure-Activity Relationship (QSAR) Models
QSAR models are mathematical constructs that establish relationships between a chemical's molecular descriptors and its biological activity or physicochemical properties. These models operate on the fundamental principle that the structure of a molecule determines its activity, enabling the prediction of toxicity for untested chemicals based on their structural similarity to compounds with known toxicological profiles. The development and application of reliable QSAR models require adherence to the OECD Principles for the Validation of QSARs, which stipulate that models must have a defined endpoint, an unambiguous algorithm, a defined domain of applicability, appropriate measures of goodness-of-fit, and a mechanistic interpretation [13].
Modern QSAR implementations include platforms such as the VEGA platform, which provides multiple models for predicting toxicity endpoints, and the OPERA (Open (quantitative) structure–activity Relationship App), which offers predictions of physicochemical properties and toxicity endpoints for thousands of chemicals [82] [83]. These tools have gained regulatory acceptance in various frameworks, including REACH, biocides, cosmetics, and food contact materials regulations, demonstrating their growing importance in chemical safety assessment [13].
Interspecies Correlation Estimation (ICE) Models
ICE models are log-linear regression models that predict the sensitivity of untested species based on toxicity data from surrogate species. The technical basis of ICE models can be expressed by the equation: Log₁₀(Predicted Taxa Toxicity) = a + b × Log₁₀(Surrogate Species Toxicity), where a and b represent the intercept and slope parameters, respectively [84]. These models are developed through rigorous statistical analysis of paired toxicity data across multiple species and chemicals, requiring a minimum of three common chemicals for each species pair to establish a reliable correlation [84].
The Web-ICE platform (www3.epa.gov/webice/) represents the most comprehensive implementation of ICE models, providing models for aquatic animals, algae, and wildlife. The underlying databases are curated from high-quality sources including the US EPA ECOTOXicology Knowledgebase (ECOTOX), which contains thousands of toxicity records across diverse species [84]. Model validation is typically performed using leave-one-out cross-validation, where each data point is systematically excluded and predicted from the remaining data to evaluate predictive accuracy [84].
Table 1: ICE Model Databases in Web-ICE
| Database | Records | Species | Chemicals | Taxonomic Levels |
|---|---|---|---|---|
| Aquatic Animals | 8,632 | 316 | 1,499 | Species, Genus, Family |
| Algae | 1,647 | 69 | 457 | Species, Genus |
| Wildlife | 4,329 | 156 | 951 | Species, Family |
The Model Coupling Framework: Integrating QSAR and ICE
Conceptual Workflow for Model Integration
The integration of QSAR and ICE models follows a systematic workflow designed to maximize the strengths of each approach while mitigating their individual limitations. The coupled framework enables researchers to generate comprehensive toxicity datasets for developing robust Species Sensitivity Distributions (SSDs), which form the basis for environmental risk assessment and regulatory decision-making [85] [81].
The coupled QSAR-ICE approach begins with using QSAR models to predict toxicity for one or more standard test species based solely on the chemical's molecular structure. This initial prediction fills the critical data gap for chemicals with no available experimental toxicity data. The QSAR-predicted toxicity values then serve as input for ICE models, which extrapolate these values across a taxonomically diverse range of species. This sequential coupling effectively addresses the dual limitations of both approaches: QSAR models are typically limited to predictions for standard test species, while ICE models require toxicity data for at least one surrogate species [85] [82].
Methodological Protocol for Implementation
Phase 1: Toxicity Prediction Using QSAR Models
Chemical Structure Preparation: Obtain or draw the molecular structure of the target chemical. Represent the structure in an appropriate format such as Simplified Molecular Input Line Entry System (SMILES) or International Chemical Identifier (InChI) [83].
Model Selection: Choose appropriate QSAR models based on the endpoint of interest (e.g., acute or chronic toxicity) and the target species. Recommended platforms include:
- VEGA Hub (https://www.vegahub.eu/): Provides multiple validated models for various toxicity endpoints.
- OPERA: Offers predictions for physicochemical properties and toxicity endpoints [83].
Applicability Domain Assessment: Verify that the target chemical falls within the applicability domain of the selected QSAR models to ensure reliable predictions.
Toxicity Prediction: Execute the models to obtain predicted toxicity values (e.g., LC50, EC50, NOEC) for standard test species such as Daphnia magna, Pimephales promelas, or Oncorhynchus mykiss.
Phase 2: Species Extrapolation Using ICE Models
Platform Access: Navigate to the Web-ICE platform (www3.epa.gov/webice/) [84].
Surrogate Species Selection: Input the species for which QSAR predictions were obtained in Phase 1.
Predicted Species Selection: Select the taxonomic groups or specific species for which toxicity predictions are needed. Web-ICE allows predictions for:
- Species-level models: Most precise, based on specific species.
- Genus-level models: Applied when species-level models are unavailable.
- Family-level models: Used when neither species nor genus models exist [84].
Model Execution and Validation: Run the ICE models and review the associated uncertainty statistics, including coefficient of determination (R²) and standard error, to evaluate prediction reliability.
Phase 3: Risk Assessment Application
SSD Construction: Compile the ICE-predicted toxicity values for multiple species and fit a statistical distribution (e.g., log-normal, log-logistic) to create a Species Sensitivity Distribution [85] [81].
HC₅ Derivation: Calculate the Hazard Concentration for 5% of species (HC₅) from the SSD, representing the concentration expected to protect 95% of species [81].
PNEC Calculation: Derive the Predicted No-Effect Concentration (PNEC) by applying an appropriate assessment factor to the HC₅ or by direct use of the HC₅ value, depending on the regulatory framework [85] [82].
Risk Quotient (RQ) Determination: Calculate risk quotients by dividing measured environmental concentrations (MECs) by the PNEC. RQ values <0.1 indicate low risk, 0.1-1.0 indicate medium risk, and >1.0 indicate high risk [85] [82].
Table 2: Case Study Applications of Coupled QSAR-ICE Models
| Chemical Class | Assessment Purpose | Key Findings | Reference |
|---|---|---|---|
| Bisphenols (BPA, BPS, BPF) | Ecological risk assessment in Chinese surface waters | Derived PNECs of 8.04, 35.2, and 34.2 μg/L for BPA, BPS, and BPF, respectively; overall low ecological risk (RQ <0.1 in most cases) with some localized higher risks | [85] [82] |
| 6PPD and 6PPD-Q | Water quality criteria development for tire wear derivatives | Established short-term WQC of 26.02 μg/L (6PPD) and 0.20 μg/L (6PPD-Q); 6PPD-Q posed significant ecological risks in urban-influenced waters | [81] |
| Alkylphenol Substances | River ecological risk assessment in megacity | Predicted PNECs for multiple alkylphenol substances using QSAR-ICE-SSD approach; applied to urban river risk assessment | [81] |
| Per- and Polyfluoroalkyl Substances (PFAS) | Screening-level ecological risk assessment | Validated coupled model accuracy by comparing PNECs derived from actual toxicity data with model-based results | [82] |
Successful implementation of coupled QSAR-ICE modeling requires access to specialized computational tools, databases, and software platforms. The following table summarizes the essential "research reagents" for conducting these analyses.
Table 3: Essential Research Tools for QSAR-ICE Modeling
| Tool/Resource | Type | Primary Function | Access Information |
|---|---|---|---|
| Web-ICE | ICE Platform | Predicts toxicity to untested species using interspecies correlations | https://www.epa.gov/webice/ [84] |
| VEGA Hub | QSAR Platform | Provides multiple QSAR models for toxicity prediction | https://www.vegahub.eu/ [82] |
| ICE (Integrated Chemical Environment) | Data Repository & Tools | Curated toxicity data, physicochemical properties, and extrapolation tools | https://ice.ntp.niehs.nih.gov/ [86] [83] |
| US EPA ECOTOX | Toxicity Database | Comprehensive database of experimental toxicity values | https://cfpub.epa.gov/ecotox/ [84] [82] |
| OPERA | QSAR Tool | Predicts physicochemical properties and toxicity endpoints | Available within ICE platform [83] |
| SARA-ICE | Specialized ICE Tool | Points of departure for skin sensitization risk assessment | OECD Test Guideline 497 [87] |
Validation and Uncertainty Analysis in Coupled Models
Model Performance Assessment
Rigorous validation is essential for establishing confidence in coupled QSAR-ICE predictions. The validation process typically involves comparing model predictions with experimentally determined toxicity values not used in model development. Key performance metrics include the N-fold difference between predicted and measured values, with a common benchmark being whether predictions fall within a factor of 10 of experimental values [84]. For ICE models specifically, leave-one-out cross-validation provides a robust measure of predictive capability, where each observed data point is sequentially excluded and predicted from the remaining data [84].
In a study validating coupled models for bisphenols, researchers demonstrated the approach's reliability by comparing predicted and experimental toxicity values, finding consistent agreement across multiple species [85] [82]. Similarly, validation exercises for per- and polyfluoroalkyl substances (PFAS) showed that PNECs derived from coupled models closely matched those derived from experimental data, supporting the use of these approaches for screening-level risk assessment [82].
Uncertainty Characterization and Propagation
Understanding and quantifying uncertainty is crucial when using coupled models for regulatory decision-making. The primary sources of uncertainty in QSAR-ICE modeling include:
- QSAR Model Uncertainty: Arising from limitations in training data, applicability domain issues, and algorithmic constraints.
- ICE Model Uncertainty: Stemming from variability in interspecies relationships, taxonomic distance between surrogate and predicted species, and chemical mode of action.
- SSD Model Uncertainty: Resulting from species selection, distributional assumptions, and sample size limitations.
The Web-ICE platform provides uncertainty statistics for each prediction, including standard error and confidence intervals, enabling users to evaluate the reliability of extrapolations [84]. Furthermore, the application of assessment factors to HC₅ values when deriving PNECs incorporates an additional layer of conservatism to account for residual uncertainty [85] [81].
Regulatory Context and Future Perspectives
The application of coupled QSAR-ICE models aligns with international initiatives to promote New Approach Methodologies (NAMs) in chemical safety assessment. Regulatory frameworks including REACH, cosmetics regulation, and pesticide risk assessment increasingly encourage the use of these approaches to reduce animal testing while maintaining protection of human health and the environment [1] [13]. The European Chemicals Agency (ECHA) and the Organisation for Economic Co-operation and Development (OECD) have established guidelines for using computational approaches, including the Read-Across Assessment Framework (RAAF) and OECD QSAR Validation Principles [13].
Future developments in coupled modeling are likely to focus on several key areas:
- Integration with Adverse Outcome Pathways (AOPs): Linking chemical perturbations to biological responses across levels of organization [87].
- Enhanced ICE Models: Expanding taxonomic coverage and chemical domains to improve predictive capability.
- High-Throughput Toxicokinetics: Incorporating physiologically based pharmacokinetic (PBPK) models and in vitro to in vivo extrapolation (IVIVE) to refine toxicity predictions [86] [83].
- Machine Learning Approaches: Leveraging advanced algorithms to improve prediction accuracy and characterize uncertainty.
As these methodologies continue to evolve, coupled QSAR-ICE approaches are poised to become increasingly central to ecological risk assessment, enabling more efficient and comprehensive evaluation of chemical hazards while supporting the global transition toward animal-free toxicology testing.
In silico methods, particularly Quantitative Structure-Activity Relationship (QSAR) models, have become indispensable tools in modern environmental risk assessment (ERA). Driven by regulatory pressures to reduce animal testing and manage vast numbers of chemicals, these computational approaches support the hazard identification of substances ranging from pesticides to cosmetic ingredients [37] [1]. However, the predictive outputs of these models are not infallible, making the assessment of their reliability a cornerstone of their scientific and regulatory acceptance. Uncertainty in predictions may arise from concerns regarding the quality of training data, the appropriateness of the chemical applicability domain, and the interpretability of the relationship between input features and output [88]. This guide provides a comprehensive technical framework for researchers and assessors to systematically evaluate, quantify, and communicate the confidence in predictions generated by in silico models for environmental risk assessment, ensuring they are fit for purpose in a regulatory context.
Key Components of Predictive Reliability
The reliability of a (Q)SAR prediction is not a single value but a composite assessment based on several pillars. Understanding these components is crucial for a nuanced confidence evaluation.
Applicability Domain (AD): The AD is the chemical space defined by the model's training data and the algorithm. Predictions for chemicals falling within this domain are considered more reliable. A target chemical should be structurally similar to other chemicals used to train the model to be considered part of the AD [89]. The definition and verification of how a substance under analysis relates to the AD is a critical element for validation [89].
Quantitative vs. Qualitative Predictions: As a general rule, qualitative predictions (e.g., classifying a substance as biodegradable or not) are often more reliable than quantitative predictions (e.g., predicting an exact biodegradation half-life) when evaluated against regulatory criteria like those in REACH and CLP [37].
Model Robustness and Performance Metrics: This refers to the statistical performance of the model on validation datasets, often measured by metrics such as accuracy, sensitivity, and specificity. Different models, even for the same endpoint, can show inconsistencies in results, underscoring the need for a weight-of-evidence approach [89].
Problem Formulation as a First Step
Before running any model, a critical yet often overlooked step is Problem Formulation (PF). PF is a systematic and iterative process aimed at identifying and defining factors critical to the assessment [88]. A well-defined PF for an in silico toxicology study should:
- Characterize the scope and context of the assessment.
- Identify research needs and data gaps.
- Develop a conceptual model of the system.
- Formulate a testable hypothesis [88]. An inadequately formulated problem can lead to an inadequate specification of risk concerns or insufficient clarity regarding the applicability domain of a model, thereby introducing significant uncertainty at the outset of the assessment [88].
Table 1: Key Components of a Problem Formulation for In Silico Toxicology
| Component Category | Description | Role in Mitigating Uncertainty |
|---|---|---|
| Assessment Context & Scope | Defines the regulatory purpose, the chemical categories of interest, and the specific endpoints to be predicted. | Ensures the model is applied to an appropriate context, preventing misapplication. |
| Conceptual Model | A representation of the hypothesized causal relationships between chemical exposure and the ecological effect. | Provides a scientific rationale for model selection and helps interpret results. |
| Analysis Plan & Hypothesis | Specifies the models to be used, the data required, and the testable hypothesis about the chemical's hazard. | Creates a transparent and pre-defined workflow, reducing selective reporting bias. |
A Tiered Framework for Confidence Assessment
A tiered framework allows for a scalable and efficient assessment, starting with simpler, higher-throughput methods and progressing to more complex evaluations as needed.
Tier 1: Initial Model Evaluation and Weight of Evidence
The initial tier focuses on gathering multiple lines of evidence from readily available models.
Using Multiple Models: Do not rely on a single model. Instead, use a suite of models and integrate the generated predictions. When results from independent models align, confidence in predictions increases [89]. For instance, in predicting the persistence of cosmetic ingredients, the Ready Biodegradability IRFMN model (VEGA), Leadscope model (Danish QSAR Model), and BIOWIN model (EPISUITE) were found to show the highest performance [37].
Battery Calls and Weight of Evidence: Some platforms, like the Danish (Q)SAR software, incorporate “battery calls”—majority-based predictions where at least two out of three models agree within the applicability domain [89]. This integration of results provides a more robust overall call than any single model.
Qualitative Assessment of the Applicability Domain: Most tools provide an indication of whether the query chemical falls within the model's AD. This initial check is a fundamental first step in gauging reliability.
Table 2: Examples of High-Performing Models for Key Environmental Endpoints
| Environmental Endpoint | Recommended (Q)SAR Models/Tools | Type of Prediction |
|---|---|---|
| Persistence (Ready Biodegradability) | Ready Biodegradability IRFMN (VEGA), Leadscope (Danish QSAR), BIOWIN (EPISUITE) | Qualitative & Quantitative [37] |
| Bioaccumulation (Log Kow) | ALogP (VEGA), ADMETLab 3.0, KOWWIN (EPISUITE) | Quantitative [37] |
| Bioaccumulation (BCF) | Arnot-Gobas (VEGA), KNN-Read Across (VEGA) | Quantitative [37] |
| Mobility (Log Koc) | OPERA v. 1.0.1 (VEGA), KOCWIN-Log Kow (VEGA) | Quantitative [37] |
Tier 2: Quantitative and Statistical Integration
If a higher degree of confidence is required, a more rigorous statistical analysis of the predictions is warranted.
Principal Component Analysis (PCA) and Cluster Analysis: These techniques can be applied to evaluate the concordance among different predictive models. They help visualize and assess the similarity in both the predictions and the chemical space covered by different models, elucidating the relationship between AD overlapping and prediction concordance [89].
Correlation Analysis: Investigating the correlation between predictions from different models can reveal redundancy. Highly correlated models do not add new information, whereas independent models that agree provide a stronger weight of evidence [89].
The workflow for a tiered confidence assessment, from problem formulation to final decision, is outlined below.
Tier 3: Integration with New Approach Methodologies (NAMs)
For the highest level of confidence, especially in regulatory decision-making for complex endpoints, in silico predictions should be integrated with other New Approach Methodologies (NAMs) in a Next-Generation Risk Assessment (NGRA) framework [90].
Hypothesis-driven Hazard Identification: Using high-throughput screening data (e.g., from ToxCast) to establish bioactivity indicators for specific genes and tissues, which can be used to form a hypothesis that in silico models can help test [90].
Toxicokinetic (TK) Modeling: TK models can be used to estimate internal concentrations at the target site. This allows for a more relevant risk assessment by comparing modeled internal doses to bioactivity concentrations from in vitro assays, moving beyond external dose comparisons [90]. A tiered NGRA framework can refine the assessment by integrating TK modeling to compare in vitro bioactivity with in vivo outcomes and realistic exposure estimations [90].
Experimental Protocols for Key Confidence-Building Analyses
Protocol: Conducting a Multi-Model Concordance Analysis
Objective: To statistically evaluate the concordance between different (Q)SAR models applied to a common set of chemicals, assessing both prediction similarity and applicability domain overlap.
Materials:
- Chemical Dataset: A set of chemicals (e.g., pesticide-active substances and metabolites) with known experimental outcomes for the endpoint of interest (e.g., carcinogenicity) is ideal for validation [89].
- Software Tools: OECD QSAR Toolbox (v4.5 or higher) and/or the Danish (Q)SAR Software [89].
- Statistical Software: R, Python (with pandas, scikit-learn, matplotlib), or similar for multivariate analysis.
Methodology:
- Data Matrix Construction: For each chemical in the dataset, export prediction results and applicability domain information from multiple models within your chosen software (e.g., the Danish (Q)SAR database) [89].
- Data Preprocessing: Encode categorical predictions (e.g., Positive, Negative) numerically. Create a matrix where rows are chemicals and columns are the different model predictions.
- Principal Component Analysis (PCA): Apply PCA to the data matrix to reduce dimensionality and visualize the clustering of chemicals based on their profile of predictions across all models. This helps identify outliers and patterns of consensus or disagreement [89].
- Cluster Analysis: Perform hierarchical clustering on the prediction data to group chemicals with similar prediction profiles and to group models that yield similar results.
- Correlation Analysis: Calculate correlation coefficients (e.g., Pearson or Spearman) between the continuous outputs or ranked predictions of different models. This quantifies the redundancy between models.
- Interpretation: A high concordance between models, evidenced by tight clustering in PCA and high correlation coefficients, increases overall confidence. Disparate results indicate a need for a more cautious, weight-of-evidence approach and may necessitate moving to a higher tier of assessment.
Protocol: Implementing a Tiered NGRA with TK Modeling
Objective: To refine the risk assessment of a chemical (e.g., a pyrethroid pesticide) by integrating in silico and in vitro bioactivity data with toxicokinetic modeling to compare external and internal doses.
Materials:
- Bioactivity Data: AC50 values from in vitro high-throughput screening assays (e.g., ToxCast database) [90].
- TK Model: A physiologically based toxicokinetic (PBTK) model suitable for the chemical class and species of interest (e.g., generic avian or mammalian models) [2] [90].
- Exposure Data: Realistic exposure estimations (e.g., from human biomonitoring or food residue monitoring) [90].
Methodology:
- Tier 1 - Bioactivity Data Gathering: Obtain in vitro bioactivity data (AC50 values) for the chemical(s) of interest. Categorize data by relevant biological pathways or tissue systems [90].
- Tier 2 - Combined Risk Assessment: Calculate relative potencies based on in vitro AC50s and compare them to relative potencies derived from traditional points of departure like No-Observed-Adverse-Effect Levels (NOAELs) or Acceptable Daily Intakes (ADIs). This tests the hypothesis of a common mode of action [90].
- Tier 3 - TK-Mediated Margin of Exposure (MoE) Analysis:
- Use the TK model to estimate the internal concentration (e.g., in plasma or tissue) that would result from the in vivo NOAEL (the bioactivity equivalent dose).
- Similarly, use the TK model to estimate the internal concentration resulting from realistic human exposure levels.
- Calculate a bioactivity-adjusted MoE as:
MoE = (Internal concentration at NOAEL) / (Internal concentration at human exposure).
- Interpretation: This refined MoE, based on internal dose, provides a more biologically relevant risk estimate than an MoE based on external dose alone, thereby increasing confidence in the final risk conclusion [90].
The Scientist's Toolkit: Essential Research Reagents and Platforms
Table 3: Key Software and Databases for In Silico Confidence Assessment
| Tool Name | Type | Primary Function in Confidence Assessment |
|---|---|---|
| Danish (Q)SAR Software | Software Platform | Provides a database of predictions from >200 models and "battery calls" for weight-of-evidence assessment [89]. |
| OECD QSAR Toolbox | Software Platform | Supports grouping, read-across, and (Q)SAR, providing multiple methods to fill data gaps and assess consistency [89]. |
| VEGA | Software Platform | Suite of (Q)SAR models for fate and toxicity; models often include transparent applicability domain assessments [37]. |
| EPI Suite | Software Platform | Provides a suite of models for predicting physical/chemical properties and environmental fate parameters [37]. |
| ToxCast Database | Database | Source of high-throughput in vitro bioactivity data (AC50s) for hypothesis generation and integration with in silico predictions [90]. |
| ADMETLab 3.0 | Software Platform | Used for predicting key properties like Log Kow, contributing to bioaccumulation assessment [37]. |
The reliability of in silico predictions in environmental risk assessment is not a binary outcome but a continuum that must be actively evaluated and communicated. A systematic approach—beginning with a clear problem formulation, progressing through a tiered framework of multi-model and statistical checks, and culminating in integration with other NAMs—provides a robust and defensible pathway to confidence. As the field evolves, the development of standardized and transparent frameworks for assessing uncertainty will be paramount to increasing the regulatory acceptance and scientific impact of these powerful in silico tools, ultimately supporting the safety assessment of chemicals in our environment without relying on animal testing.
In silico environmental risk assessment (ERA) has emerged as a fundamental methodology for evaluating the potential impact of chemicals on ecosystems, leveraging computational approaches to predict fate, transport, and toxicity. Traditional ERA typically focuses on individual chemical entities, examining them in isolation to determine safe exposure levels for organisms and ecosystems. However, this approach fails to capture the reality of environmental exposure, where organisms are invariably subjected to complex mixtures of chemicals that may interact, leading to combined effects that differ from those predicted from single-substance assessments [10] [2]. The challenge of mixtures and multiple stressors represents one of the most significant frontiers in toxicology and environmental science, demanding innovative computational strategies to move from reductionist to holistic assessment paradigms.
The production volume of synthetic chemicals continues to increase globally, expected to triple by 2050 compared to 2010 levels [91]. This expansion, coupled with the identification of countless transformation products and metabolites, creates exposure scenarios of immense complexity. Most real-world exposures involve complex mixtures of chemicals found in food, beverages, the environment, and consumer products [10]. Assessing these mixtures presents extraordinary challenges: the number of possible combinations is virtually infinite, chemical components may interact synergistically or antagonistically, and their identities and proportions may be unknown or variable. In silico methods provide the only practical approach to begin addressing this complexity, offering tools to prioritize mixtures for testing, predict potential interactions, and extrapolate from limited experimental data.
Current Framework and Limitations in Mixture Assessment
The Traditional Risk Assessment Paradigm
The established framework for environmental risk assessment follows a well-defined sequence of steps, typically including hazard identification, exposure assessment, toxicity assessment, and risk characterization [2] [91]. For individual chemicals, this process has been refined over decades and incorporates increasingly sophisticated computational tools. The quantitative risk assessment methodology combines knowledge about the overall frequency of health parameters in specific populations, the distribution of exposures, and dose-response functions typically derived from epidemiological or toxicological studies [91]. This approach answers questions such as "How many disease cases are attributable to this exposure factor?" or "How many disease cases would be avoided if this policy was implemented?"
Specific Challenges in Mixture Toxicology
The application of this established framework to chemical mixtures encounters several specific scientific and methodological challenges:
- Identification Complexity: Comprehensive analytical characterization of all components in environmental mixtures is resource-intensive, and many components may be present at levels below analytical detection limits yet still contribute to biological activity.
- Interaction Potential: Chemicals in mixtures may interact through various mechanisms, including receptor competition, metabolic interference, or signaling pathway cross-talk, leading to non-additive effects.
- Temporal Dynamics: Components may have different environmental fates and persistence, causing the mixture composition to change over time and space.
- Threshold Considerations: The traditional threshold of toxicological concern approach becomes complicated when applied to mixtures with multiple components, each potentially below individual thresholds but collectively active.
In Silico Tools for Mixture Exposure and Toxicity Assessment
Computational Approaches for Exposure Assessment
Exposure modeling for mixtures requires predicting the co-occurrence of multiple chemicals in environmental compartments and biota. Advanced tools have been adapted or developed specifically for this purpose.
Table 1: Computational Tools for Assessing Pesticide Exposure in Environmental Mixtures
| Tool Name | Application Scope | Methodology | Key Outputs |
|---|---|---|---|
| AGDISP | Pesticide spray drift into air systems | Lagrangian particle model | Deposition and drift estimates up to 400m from application site [2] |
| TOXSWA | Pesticide fate in water, sediment, and macrophytes | Process-based simulation | Concentrations in water bodies over time [2] |
| SWAT | Watershed-scale pesticide loading | Hydrologic transport modeling | Pesticide fluxes into river systems [2] |
These exposure models incorporate spatial and temporal dimensions to predict chemical concentrations in various environmental media. When applied to mixtures, they can be run in parallel for multiple chemicals to identify scenarios of co-occurrence that merit further investigation for potential interactive effects.
Predictive Approaches for Mixture Toxicity
Toxicity prediction for mixtures employs both mechanism-based and statistical approaches. The field has evolved from simple concentration addition models to more sophisticated methods that incorporate chemical structures and biological pathways.
Table 2: In Silico Models for Toxicity Assessment of Chemical Mixtures
| Model Name | Organism/Focus | Methodology | Performance Metrics |
|---|---|---|---|
| BeeTox | Honeybee toxicity | Graph Attention Convolutional Neural Network (GACNN) | Accuracy: 0.837, Specificity: 0.891, Sensitivity: 0.698 [2] |
| QSAR for Metabolites | Toxicity of transformation products | Quantitative Structure-Activity Relationships | Varies by endpoint; generally >0.7 accuracy for major endpoints [10] |
| GACNN for Binary Mixtures | Honey bee toxicity of organic mixtures | Innovative QSAR modeling | Capable of distinguishing bee-toxic chemical combinations [2] |
These models demonstrate that computational approaches can successfully predict toxicity even for complex scenarios involving multiple chemicals. The BeeTox model, in particular, represents a significant advancement through its application of graph neural networks to capture structural features relevant to toxicity mechanisms.
Methodological Protocols for Mixture Assessment
Standardized In Silico Toxicology Protocols
The development of standardized in silico toxicology protocols ensures that assessments are performed and evaluated consistently across different institutions and regulatory bodies [10]. For mixture assessment, these protocols should encompass:
1. Component Identification and Prioritization
- Utilize analytical chemistry data to identify mixture components above established Analytical Evaluation Thresholds (AET) [10]
- Apply prioritization algorithms based on abundance, persistence, bioaccumulation potential, and structural alerts for toxicity
- Employ cheminformatics tools to cluster components by structural similarity and predicted mechanism of action
2. Interaction Potential Assessment
- Apply molecular docking and binding prediction to identify potential receptor competition
- Use metabolic network modeling to predict interference with detoxification pathways
- Implement systems biology approaches to identify pathway interactions and potential synergistic nodes
3. Combined Effect Prediction
- Employ mixture QSAR models that incorporate descriptors for multiple chemicals
- Implement hierarchical models that first predict individual chemical effects then modify based on interaction potentials
- Apply machine learning approaches trained on existing mixture toxicity data
4. Uncertainty Quantification
- Propagate uncertainties from individual chemical predictions through the mixture models
- Employ probabilistic modeling to characterize confidence in interaction predictions
- Conduct sensitivity analyses to identify the most influential parameters affecting mixture risk estimates
Workflow for Integrated Mixture Risk Assessment
The following diagram illustrates the comprehensive workflow for assessing mixtures and multiple stressors using in silico approaches:
In Silico Mixture Assessment Workflow
This workflow systematically progresses from mixture characterization through to risk quantification, with explicit consideration of interactions and uncertainties at each stage.
Implementing robust mixture assessment requires specialized computational tools and resources. The following table catalogs essential components of the mixture toxicologist's toolkit.
Table 3: Research Reagent Solutions for In Silico Mixture Assessment
| Tool Category | Specific Tools/Resources | Function in Mixture Assessment |
|---|---|---|
| Chemical Structure Databases | EPA CompTox Chemicals Dashboard, PubChem | Identify mixture components and retrieve structural information for QSAR modeling [10] |
| Toxicity Prediction Platforms | OECD QSAR Toolbox, VEGA, TEST | Predict baseline toxicity of individual mixture components using (Q)SAR approaches [10] [2] |
| Exposure Modeling Software | AGDISP, TOXSWA, SWAT | Predict environmental co-occurrence of multiple chemicals [2] |
| Metabolite Prediction Tools | Meteor, BioTransformer | Identify potential biotransformation products that may contribute to mixture effects [10] |
| Pathway Analysis Resources | KEGG, Reactome, WikiPathways | Map potential interactions between chemicals through shared biological targets [2] |
| Mixture Toxicity Databases | ACToR, CEBS | Access experimental data on chemical mixtures for model training and validation [10] |
These resources enable researchers to address the key challenges in mixture assessment, from initial component identification to final risk characterization. The integration of these tools into coherent workflows represents the state of the art in mixture risk assessment.
Signaling Pathways in Mixture Toxicology
Understanding the molecular mechanisms through which chemical mixtures exert their effects requires mapping their interactions with biological signaling pathways. The following diagram illustrates key pathways frequently affected by chemical mixtures and their potential points of interaction.
Key Pathways in Mixture Toxicology
These pathways represent critical targets for chemical mixtures and illustrate how components might interact through shared or interconnected biological processes. The nuclear receptor pathways are particularly vulnerable to mixture effects due to the potential for multiple chemicals to act as agonists, antagonists, or synergists at the same receptor.
Future Directions and Research Needs
The field of mixture toxicology stands at a pivotal point, with several promising avenues for advancement:
1. High-Throughput Transcriptomics and Bioinformatics
- Development of pattern-based mixture assessment using gene expression signatures
- Application of deep learning to identify characteristic response patterns for mixture types
- Integration of toxicogenomics data across multiple chemicals to predict mixture effects
2. Advanced Interaction Modeling
- Implementation of network pharmacology approaches to predict polypharmacology effects
- Development of dynamic models that capture temporal aspects of mixture exposures
- Creation of adverse outcome pathway networks for complex mixtures
3. Integrated Testing Strategies
- Combination of in silico, in vitro, and limited in vivo data in weight-of-evidence frameworks
- Development of tiered testing strategies that use computational approaches to guide empirical testing
- Implementation of bioassay-directed fractionation coupled with computational identification
4. Regulatory Adoption Frameworks
- Establishment of standardized protocols for regulatory acceptance of mixture assessments [10]
- Development of transparent validation frameworks for mixture models
- Creation of regulatory decision trees for different mixture scenarios
The successful application of in silico tools for mixture risk assessment has demonstrated substantial benefits, including the potential to eliminate 100,000-150,000 test animals and save $50,000,000-$70,000,000 for assessing 261 substances [92]. As these methodologies continue to evolve, they promise to transform our approach to chemical mixture assessment, enabling more protective and predictive safety evaluation while reducing animal testing and associated costs.
Addressing mixtures and multiple stressors represents a critical frontier in environmental risk assessment—one that demands a fundamental shift from single-chemical to holistic approaches. In silico methods provide the only feasible foundation for this paradigm shift, offering the scalability, mechanistic insight, and integrative capacity needed to navigate the complexity of real-world exposure scenarios. While significant challenges remain, particularly in predicting non-additive interactions and validating integrated approaches, the rapid advancement of computational toxicology holds immense promise. Through continued development of standardized protocols, innovative modeling techniques, and collaborative frameworks for data sharing and validation, the field is poised to transform mixture assessment from an intractable challenge to a manageable component of comprehensive environmental protection.
Benchmarking Success: Validating Models and Comparing In Silico vs. Conventional ERA
In silico models, which use computational simulations to predict the behavior and effects of chemicals, are becoming indispensable tools in modern environmental risk assessment (ERA). These models include Quantitative Structure-Activity Relationship (QSAR) models for predicting chemical properties and toxicity, toxicokinetic-toxicodynamic (TK-TD) models that simulate the internal dose and biological effects of chemicals over time, and dynamic energy budget (DEB) models that assess chemical impacts on organism growth and reproduction. The credibility of these models, however, hinges on rigorous validation against experimental data to ensure their predictions are reliable for regulatory decision-making. As regulatory agencies increasingly accept computational evidence, establishing robust validation paradigms has become critical for ensuring these models produce trustworthy results for environmental safety evaluations [1] [93].
The validation process is particularly crucial for implementing the tiered approaches common in environmental risk assessment. In these frameworks, initial conservative models with limited data requirements are used for screening, followed by more complex models with higher data demands for refined assessments. Without proper validation, the predictions that guide these tiered decisions could lead to either unnecessary over-protection or dangerous under-protection of ecosystems [1]. Recent advancements have seen regulatory agencies such as the FDA and EMA beginning to accept in silico evidence as part of submissions, particularly when models undergo comprehensive verification, validation, and uncertainty quantification [93] [94].
Fundamental Principles of Model Verification and Validation
Distinguishing Verification and Validation
In the context of in silico models, verification and validation represent distinct but complementary processes. Verification addresses the question "Did we build the model correctly?" while validation addresses "Did we build the correct model?" Specifically, verification ensures the computational implementation accurately represents the intended mathematical model, whereas validation determines how well the model's predictions correspond to real-world observations [94].
The context of use (COU) is a fundamental concept that guides the validation process. The COU explicitly defines the specific regulatory purpose and applicability of the model, including the specific endpoints predicted, the chemical classes covered, and the environmental scenarios addressed. The validation requirements for a model depend directly on its COU—models supporting critical regulatory decisions with significant potential risk require more extensive validation than those used for preliminary screening [94].
The ASME V&V-40 Standard Framework
The ASME V&V-40 technical standard provides a methodological framework for assessing the credibility of computational models used in regulatory evaluations of biomedical products. This framework has been adapted for environmental risk assessment applications and involves a structured process [94]:
- Define Context of Use: Precisely specify the model's purpose, including the specific risk assessment questions it will address, the types of chemicals it will evaluate, and the environmental conditions under which it applies.
- Risk Analysis: Identify potential impact of incorrect model predictions to determine the necessary level of validation evidence.
- Credibility Assessment: Evaluate the model through verification, validation, and uncertainty quantification activities.
- Credibility Decision: Determine whether the model has sufficient credibility for its specified COU.
This framework emphasizes that model credibility exists on a spectrum—it is not a binary state—and should be commensurate with the model's potential impact on regulatory decisions and environmental protection [94].
Technical Approaches to Model Validation
Quantitative Validation Metrics and Statistical Measures
Rigorous validation requires quantitative metrics to compare model predictions with experimental data. The following table summarizes key statistical measures used in validation studies:
Table 1: Key Statistical Metrics for Model Validation
| Metric | Calculation | Interpretation | Ideal Value | ||
|---|---|---|---|---|---|
| Coefficient of Determination (R²) | 1 - (SS₍ᵣₑₛ₎/SS₍ₜₒₜ₎) | Proportion of variance explained by model | Close to 1.0 | ||
| Root Mean Square Error (RMSE) | √(Σ(Pᵢ - Oᵢ)²/n) | Average magnitude of prediction error | Close to 0 | ||
| Mean Absolute Error (MAE) | Σ | Pᵢ - Oᵢ | /n | Average absolute difference | Close to 0 |
| Concordance Correlation Coefficient (CCC) | (2rσₚσₒ)/(σₚ² + σₒ² + (μₚ - μₒ)²) | Agreement between predicted and observed | Close to 1.0 |
These metrics evaluate different aspects of model performance. For example, Q² (cross-validated R²) is particularly important for QSAR models to assess predictive ability for new chemicals not used in model development. A Q² > 0.6 is generally considered acceptable for predictive models [95] [96].
Validation Workflows and Experimental Protocols
A standardized validation workflow ensures consistent evaluation of in silico models. The following diagram illustrates a comprehensive validation process adapted from regulatory guidance:
Diagram 1: Model Validation Workflow
The experimental protocols for generating validation data vary by model type but share common elements. For QSAR models predicting ecotoxicity endpoints, standard OECD test guidelines provide the experimental basis for validation:
- Algae growth inhibition tests (OECD 201): 72-hour exposure assessing effects on growth rate
- Daphnia reproduction tests (OECD 211): 21-day exposure measuring survival and reproduction
- Fish early life stage tests (OECD 210): Embryo-larval exposure assessing development and survival
These standardized tests generate the experimental data used to validate model predictions of chemical effects on aquatic organisms [97]. For PBT assessments (Persistence, Bioaccumulation, and Toxicity), validation data comes from:
- Biodegradation tests (OECD 301): Measuring biological degradation over 28 days
- Bioaccumulation tests (OECD 305): Determining bioconcentration factors in fish
- Chronic toxicity tests: As described above for toxicity assessment [97]
The quality of experimental data used for validation significantly impacts the credibility of the validation exercise. Regulatory agencies recommend using reliable data generated according to standardized test guidelines, preferably following Good Laboratory Practice (GLP) principles. Data from literature sources should undergo reliability assessment using established criteria such as the Klimisch score [98].
Case Studies in Environmental Risk Assessment
QSAR Validation for Tattoo Ink Safety Assessment
A recent comprehensive study demonstrated the application of validation paradigms for QSAR models assessing genotoxicity of tattoo ink ingredients. Researchers developed and validated custom QSAR models specifically for tattoo ink components using the following methodology [95]:
- Data compilation: Collected experimental in vitro genotoxicity data from the IUCLID database for 180 tattoo ink substances
- Model development: Built structural alerts and statistical QSAR models using curated chemical structures
- Validation protocol:
- Internal validation: 5-fold cross-validation with Q² > 0.7
- External validation: Hold-out test set with concordance > 80%
- Applicability domain assessment: Defined chemical space where reliable predictions expected
The validation results demonstrated 85% concordance with experimental Ames test data within the models' applicability domain, providing sufficient confidence to prioritize substances for further testing. This approach allowed identification of 4 high-priority, 18 medium-priority, and 2 low-priority substances from the tattoo ink ingredient list [95].
Mechanistically Validated Models for Regulatory Acceptance
Beyond statistical validation, establishing mechanistic validity significantly increases regulatory acceptance of in silico models. The mechanism of action (MechoA) approach determines whether a QSAR model is applicable to the substance under review based on its presumed biological activity [96].
For example, models validated specifically for non-polar narcotics (baseline toxicity) should not be applied to reactive chemicals without demonstrating the model captures the relevant reaction chemistry. This mechanistic validation involves:
- Identifying the toxicological mechanism through experimental studies or existing literature
- Verifying the model structure incorporates this mechanism (e.g., specific structural alerts for reactivity)
- Validating predictions against experimental data for chemicals with the same mechanism
This approach was successfully applied in environmental hazard assessment, where mechanistically validated QSAR models provided accurate predictions for tests that would be technically challenging to conduct in the laboratory [96].
Implementing Validation Frameworks for Regulatory Compliance
Credibility Assessment for Regulatory Submission
For in silico models used in regulatory submissions to agencies such as the EMA or FDA, a systematic credibility assessment is essential. This assessment evaluates multiple aspects of model development and validation [94]:
Table 2: Model Credibility Factors for Regulatory Submissions
| Credibility Factor | Assessment Criteria | Documentation Requirements |
|---|---|---|
| Model Development | Scientific rationale, mathematical basis, parameter estimation | Model formulation documentation, parameter sources |
| Verification | Code verification, numerical accuracy | Software testing results, mesh convergence studies |
| Validation | Comparison with experimental data, predictive performance | Validation dataset description, statistical measures |
| Uncertainty Quantification | Parameter uncertainty, model form uncertainty | Sensitivity analysis, uncertainty propagation |
| Applicability | Domain of applicability, limitations | Chemical space definition, extrapolation boundaries |
This comprehensive assessment ensures the model is appropriate for its specific regulatory context, whether for screening-level assessments or definitive risk characterizations [94].
Addressing Validation Challenges and Limitations
Several challenges persist in validating in silico models for environmental risk assessment:
- Data quality and availability: High-quality experimental data for validation may be limited, particularly for sensitive species or chronic endpoints
- Extrapolation uncertainty: Models validated for certain chemical classes may perform poorly when applied to new structural domains
- Ecological complexity: Simplified laboratory validation systems may not fully represent complex environmental exposures
Strategies to address these challenges include:
- Developing tailored QSAR models for specific regulatory endpoints rather than relying on general models
- Implementing applicability domain assessments to identify when models are being used beyond their validated scope
- Using integrated testing strategies that combine multiple in silico approaches with limited experimental data to increase confidence [95] [96]
Essential Research Tools and Reagent Solutions
The successful development and validation of in silico models requires specialized computational tools and data resources. The following table outlines key resources used in the field:
Table 3: Essential Research Tools for In Silico Model Development and Validation
| Tool Category | Specific Examples | Primary Function | Regulatory Relevance |
|---|---|---|---|
| QSAR Platforms | VEGA, OECD QSAR Toolbox, EPA TEST | Toxicity prediction, chemical categorization | Accepted for data gap filling under REACH |
| Structural Alert Tools | OECD SAR Suite, Toxtree | Identification of potentially hazardous substructures | Screening for genotoxicity alerts |
| Toxicogenomics Databases | Comparative Toxicogenomics Database, ToxCast | Mechanistic data for model validation | Mode of action analysis |
| Experimental Data Repositories | IUCLID, ECOTOX, PubChem | Source of validation data | Reliability assessment reference |
| Curated Chemical Databases | DSSTox, ChEMBL | High-quality structure information | QSAR model development |
These tools form the foundation for developing, applying, and validating in silico models for environmental risk assessment. The DSSTox database, for example, provides curated chemical structures that ensure correctness in chemical representations—a critical requirement for developing reliable QSAR models [95].
Future Directions in Model Validation
The field of in silico model validation is rapidly evolving, with several promising developments emerging:
- AI and machine learning approaches: Advanced neural network architectures are being validated for predicting complex toxicity endpoints, though these require particular attention to explainability and mechanistic interpretation [93] [99]
- Digital twin technology: Creating virtual representations of environmental systems enables validation at ecosystem levels rather than single species [93]
- Integrated validation frameworks: Developing standardized protocols that combine multiple lines of evidence for comprehensive model evaluation
As these technologies mature, validation paradigms will need to adapt to address new challenges in model transparency, reproducibility, and uncertainty characterization. The establishment of shared digital twin libraries and public-private consortia for validation benchmarking will be critical for advancing the field [93].
The following diagram illustrates the integrated future of model development and validation:
Diagram 2: Future Integrated Validation Approach
Robust validation paradigms are fundamental to establishing the scientific credibility and regulatory acceptance of in silico models in environmental risk assessment. By implementing systematic approaches that include rigorous statistical validation, mechanistic verification, and comprehensive uncertainty quantification, researchers can develop computational tools that reliably support environmental safety decisions. As regulatory agencies increasingly recognize the value of these approaches, standardized validation frameworks will become even more critical for ensuring that in silico models fulfill their potential to enhance chemical safety assessment while reducing animal testing and accelerating the development of safer chemicals and pharmaceuticals.
In silico methods, which include computational modeling, quantitative structure-activity relationships (QSARs), and other bioinformatics approaches, represent a transformative shift in environmental risk assessment (ERA) and chemical safety science [100]. These New Approach Methodologies (NAMs) are defined by their ability to deliver more protective and relevant models that have a reduced reliance on animals, while also offering significant economic and temporal advantages [100]. The transition from traditional, animal-heavy testing paradigms to data-driven computational approaches is driven by an ethical imperative aligned with the 3Rs (Replacement, Reduction, and Refinement of animal research) and a practical need for more efficient, scalable, and human-relevant safety assessments [101] [100]. This technical guide quantifies the substantial benefits of in silico methodologies across three critical dimensions: the reduction of animal use, the acceleration of testing timelines, and the decrease in associated costs, providing researchers and drug development professionals with evidence-based validation for their integration into regulatory and industrial frameworks.
Quantitative Advantages of In Silico Methods
The adoption of in silico methods offers demonstrable and significant advantages over traditional toxicity testing. The tables below synthesize specific quantitative data from the literature, providing a clear comparison of the efficiency gains.
Table 1: Overall Quantitative Reductions Offered by In Silico Methods
| Metric | Traditional Approach | In Silico Approach | Quantitative Reduction | Reference/Context |
|---|---|---|---|---|
| Animal Use | Required for most toxicity tests | Largely replaced or supplemented | Potential to eliminate 0.1–0.15 million test animals for 261 compounds | [2] |
| Cost | High (e.g., up to ~$9.9M overall for conventional pesticide testing) | Significantly lower | Potential savings of $50–70 billion for 261 compounds | [2] [12] |
| Testing Time | Chronic toxicity studies can take up to 2 years | Rapid, high-throughput screening | Time reduced to days or weeks for large compound libraries | [2] |
Table 2: Specific In Silico Tools and Their Applications in ERA
| Tool Name | Type | Application in ERA | Key Advantage |
|---|---|---|---|
| ECOSAR | Quantitative Structure-Activity Relationship (QSAR) | Predicts aquatic toxicity for fish, daphnids, and algae | Provides rapid toxicity predictions for data-poor chemicals, used by USEPA since the 1980s [12] |
| AGDISP | Exposure Model | Predicts pesticide spray drift and deposition in air | Effectively monitors drift, e.g., of atrazine up to 400m from application site [2] |
| BeeTox | Graph Attention Convolutional Neural Network (GACNN) | Assesses pesticide toxicity to honeybees | High prediction accuracy (0.837) for identifying bee-toxic chemicals [2] |
| Virtual Control Groups (VCG) | Historical Data Modeling | Replaces concurrent animal control groups in studies | Significantly reduces the number of animals used per study by leveraging curated historical data [101] |
Detailed Experimental Protocols and Methodologies
Protocol 1: Application of QSAR and ToxCast for Ecological Hazard Prioritization
This protocol outlines a methodology for comparing Points of Departure (PODs) from in silico and in vitro NAMs with in vivo ecological toxicity data to determine their utility for chemical screening and prioritization [12].
- Chemical Space Definition: Begin by identifying overlapping chemicals between large bioactivity repositories (e.g., ToxCast) and ecological toxicity databases (e.g., ECOTOX Knowledgebase). In a cited study, this process started with 4,584 ToxCast chemicals and 12,425 ECOTOX chemicals, resulting in 2,494 overlaps [12].
- Data Filtering and Curation: Apply stringent exclusionary criteria to the in vivo data to ensure quality and standardization. This includes filtering ECOTOX records based on specific data quality rules. The example study refined the 2,494 chemicals down to 649 suitable for final analysis [12].
- Derivation of Points of Departure (PODs):
- In vivo PODs: Extract the most sensitive toxicity values (e.g., LC50, EC50) from the filtered ECOTOX records for each chemical.
- In silico PODs: Calculate PODs using QSAR models like ECOSAR.
- In vitro PODs: Derive PODs from high-throughput screening data (e.g., ToxCast). Two common metrics are:
- ACC5: The 5th centile of the Activity Concentration at Cutoff.
- LCB: The Lower-bound Cytotoxic Burst, a more protective estimate based on cytotoxicity [12].
- Statistical Comparison and Correlation Analysis: Perform correlation analyses (e.g., linear regression) between the PODs derived from the different sources (ECOTOX, QSAR, ToxCast) for the entire dataset.
- Stratified Analysis by Chemical Use and Endpoint: Group the chemicals by use classes (e.g., pesticides, antimicrobials, pharmaceuticals) and by in vivo endpoint type (e.g., apical vs. biochemical, acute vs. chronic). Re-run correlation analyses within these subgroups to identify contexts where NAM-derived PODs show the strongest agreement with in vivo data [12].
Protocol 2: Implementing a Virtual Control Group (VCG)
This protocol describes the methodology for replacing concurrent animal control groups with VCGs, a powerful reduction strategy pioneered by initiatives like the IHI VICT3R project [101].
- Historical Data Acquisition and Standardization: Collect and curate historical control data (HCD) from previous, similar animal studies. The data must be stored in a standardized format, such as the ALURES database provided by the European Commission, to ensure consistency and usability [101].
- Data Curation and Adequate Matching: Rigorously curate the HCD to remove erroneous entries. For a new study, carefully match the experimental parameters (e.g., animal strain, age, sex, housing conditions, laboratory environment) to a subset of the historical data to generate a context-appropriate VCG [101].
- Statistical Generation of the VCG: Use statistical models to generate the virtual control group from the matched historical data. This process typically involves calculating the distribution of outcomes (e.g., mean, variance) for the control group based on the aggregated historical records.
- Regulatory Qualification and Integration: The VCG methodology requires qualification by regulatory bodies to be accepted for decision-making. This involves demonstrating that the VCG provides a reliable and scientifically rigorous baseline that does not compromise the validity or safety conclusions of the study [101].
Visualization of Workflows and Relationships
The following diagrams, generated with Graphviz using the specified color palette, illustrate the core workflows and logical relationships in in silico environmental risk assessment.
Tiered In Silico Risk Assessment Workflow
Virtual Control Group Implementation Logic
The successful implementation of in silico environmental risk assessment relies on a suite of computational tools, databases, and models. The following table details the essential components of this toolkit.
Table 3: Essential Research Reagents and Resources for In Silico ERA
| Tool/Resource Name | Type | Function and Application |
|---|---|---|
| ECOTOX Knowledgebase | Database | A curated repository of in vivo ecological toxicity data for over 12,000 chemicals and species, used to validate in silico models and derive traditional toxicity benchmarks [12]. |
| US EPA CompTox Chemicals Dashboard | Database | Provides access to a wealth of physicochemical, fate, and bioactivity data for thousands of chemicals, supporting QSAR modeling and exposure assessment [12]. |
| ToxCast/Tox21 | Bioactivity Database | A high-throughput screening database containing bioactivity profiles for thousands of chemicals across hundreds of assay endpoints, used to derive in vitro points of departure [12]. |
| ECOSAR | QSAR Model | A software program that uses quantitative structure-activity relationships to predict the aquatic toxicity of chemicals for which empirical test data are lacking [12]. |
| AGDISP | Exposure Model | A computational model that simulates the deposition and spray drift of pesticides applied aerially or by ground rigs, used for assessing exposure risk in air [2]. |
| ALURES Database | Database | The European Commission's database of animal test data, which serves as a key source for building historical control data (HCD) for Virtual Control Groups [101]. |
| GARDskin | In Vitro Assay | A non-animal method for assessing skin sensitization, representative of the types of NAMs used in Defined Approaches for regulatory safety assessment [100]. |
Next-Generation Risk Assessment (NGRA) represents a paradigm shift in chemical safety evaluation, moving away from traditional animal-based testing toward a hypothesis-driven, exposure-led approach. [102] This framework integrates Toxicokinetic (TK) data with New Approach Methodologies (NAMs) to enable more human-relevant, efficient, and mechanistic-based safety decisions. NGRA operates on an iterative, tiered strategy that prioritizes human biological relevance through the application of innovative tools including in vitro systems, in chemico methods, and computational (in silico) approaches. [102] [103]
The driver for adopting NGRA is multifaceted: regulatory policies such as the EU's Chemicals Strategy for Sustainability and Zero Pollution Action Plan are increasingly advocating for the Safe and Sustainable Design (SSbD) of chemicals. [102] Furthermore, the limitations of traditional animal testing in predicting human health risks, combined with ethical considerations under the 3Rs principles (Replacement, Reduction, and Refinement of animal testing), have accelerated the development and application of NAMs. [104] [105] Within environmental risk assessment research, NGRA provides a transformative framework for evaluating chemicals, pollutants, and products by leveraging advances in molecular biology, biotechnology, and computational sciences. [103]
Core Components of the NGRA Framework
Taxonomy of New Approach Methodologies (NAMs)
NAMs encompass a broad suite of innovative scientific tools that provide data on chemical hazards and exposures without relying solely on traditional animal models. [105] [103] These methodologies can be systematically categorized into several interconnected pillars, as detailed in the table below.
Table 1: Core Components of New Approach Methodologies (NAMs)
| Methodology Category | Key Technologies | Primary Applications |
|---|---|---|
| In Vitro Models [102] [105] [106] | 2D/3D cell cultures, Class="red"> [106], Organ-on-a-chip (e.g., lung-chip) [102] [105], Stem cell-based models (e.g., ReproTracker) [107] | Mechanistic toxicity screening [107], Hazard identification [106], Organ-specific toxicity assessment (e.g., developmental toxicity, cardiotoxicity) [105] [107] |
| In Chemico Methods [102] | Peptide reactivity assays (OECD 442C) [102], Methanol probe chemisorption [102] | Assessment of direct chemical reactivity, e.g., skin sensitization potential [102] |
| Computational (In Silico) Tools [102] [104] [105] | QSAR models [102] [108], Read-Across [102] [104], Physiologically Based Kinetic (PBK) models [102] [108], Artificial Intelligence/Machine Learning [105] | Toxicity prediction [104], Priority setting [108], High-throughput dose-response modeling [102], Carcinogenicity assessment (e.g., N-nitrosamines via CPCA) [104] |
| Omics Technologies [105] [106] | Transcriptomics, Proteomics, Metabolomics [105] | Uncovering toxicity pathways and mechanisms [105], Identifying biomarkers of effect [105] |
| Adverse Outcome Pathways (AOPs) [105] [107] | Structured mechanistic frameworks linking molecular initiating events to adverse outcomes [105] | Organizing knowledge for risk assessment [105], Supporting IATA (Integrated Approaches to Testing and Assessment) [107] |
The Role of Toxicokinetics (TK) in NGRA
Toxicokinetics (TK), often implemented through Physiologically Based Kinetic (PBK) models, is the cornerstone that bridges in vitro bioactivity data to in vivo human relevance. [102] TK models quantitatively describe the Absorption, Distribution, Metabolism, and Excretion (ADME) of a chemical in the body. In the NGRA context, they are crucial for performing in vitro-to-in vivo extrapolation (IVIVE). [105] This process converts effective concentrations derived from NAMs (e.g., an AC~50~ from a cell-based assay) into human equivalent external exposure doses, which can then be compared to anticipated human exposure levels to assess risk. [102] Modern high-throughput PBK models are being developed to overcome the data limitations of traditional models, enabling the application of TK principles to a wider range of chemicals in a screening context. [102]
The Tiered and Iterative NGRA Framework
The NGRA process is inherently tiered and iterative, designed to be resource-efficient and to refine hypotheses through successive stages of evaluation. [103] The workflow typically progresses from high-throughput, conservative screening to more complex, mechanistic investigations only as needed.
Figure 1: The Tiered and Iterative Workflow of Next-Generation Risk Assessment (NGRA). This diagram illustrates the stepwise process, from problem formulation to risk characterization, highlighting the iterative integration of NAM and TK data at each tier. The process allows for early exit if no risk is identified.
- Problem Formulation and Exposure Assessment: The process begins by defining the assessment goal and collating all available exposure information, including anticipated use scenarios and human exposure levels. [102] This exposure-led approach ensures the assessment is grounded in real-world relevance from the outset.
- Tier 1: Priority Setting and Initial Screening: This tier employs high-throughput NAMs, such as QSAR predictions and high-throughput in vitro screening (e.g., ToxTracker assay for genotoxicity), to rapidly identify chemicals with potential hazardous properties. [104] [107] Computational models and chemical-specific data are used to predict toxicity endpoints. If this conservative screening indicates no cause for concern, the assessment can be concluded. If a potential hazard is flagged, the process moves to Tier 2.
- Tier 2: Mechanistic Investigation: Chemicals of concern undergo more detailed evaluation using advanced in vitro models and mechanistic assays. This includes using organ-on-a-chip systems to mimic human physiology, 'omics' technologies to elucidate modes of action, and assays like ReproTracker for specific endpoints such as developmental toxicity. [102] [105] [107] The data generated here is structured within Adverse Outcome Pathway (AOP) frameworks to establish biological plausibility.
- Tier 3: Quantitative In Vitro to In Vivo Extrapolation (IVIVE): For risks that cannot be ruled out in Tiers 1 and 2, PBK models are deployed to perform quantitative IVIVE. [102] These models translate the bioactive concentrations from NAMs into human equivalent external doses. A comparison of this derived dose with the known or anticipated human exposure level forms the basis for a definitive risk characterization.
Detailed Experimental Protocols for Key NAMs
Protocol 1: ToxTracker Assay for Genotoxicity Mechanism
The ToxTracker assay is a high-throughput, mechanism-based in vitro tool used to discriminate between genotoxic carcinogens and non-carcinogens by identifying specific toxicity pathways. [107]
- 1. Principle: The assay employs mouse embryonic stem (mES) cells containing green fluorescent protein (GFP) reporters fused to genes activated by specific cellular stress pathways, including DNA damage (Rtkn-GFP for strand breaks, Bscl2-GFP for oxidative stress), and cellular stress (Btg2-GFP for p53 activation, Ddit4-GFP for general cellular stress, Srxn1-GFP for protein damage, and Blvrb-GFP for oxidative stress). [107]
- 2. Methodology:
- Cell Culture and Seeding: Grow ToxTracker mES cells under standard conditions. Seed cells into multi-well plates and allow to adhere.
- Chemical Treatment: Expose cells to a range of concentrations of the test chemical, including a vehicle control and appropriate positive controls (e.g., methyl methanesulfonate for clastogens, acrylamide for aneugens). A typical exposure time is 24 hours.
- Flow Cytometry Analysis: Harvest the cells and analyze GFP expression using a flow cytometer. The percentage of GFP-positive cells in each reporter line is quantified.
- Cytotoxicity Assessment: Perform a concurrent cytotoxicity assay (e.g., using a membrane integrity dye) to ensure that any negative results are not due to compound toxicity.
- 3. Data Interpretation:
- Induction of Rtkn and Bscl2 indicates a clastogenic (DNA-breaking) mechanism.
- Induction of Btgl2 and Ddit4 without Rtkn or Bscl2 induction suggests a non-genotoxic mechanism or aneugenic (chromosome mis-segregation) activity.
- The specific combination of activated reporters provides a mechanistic fingerprint for the test compound, aiding in more accurate risk assessment. [107]
Protocol 2: Physiologically Based Kinetic (PBK) Modeling for IVIVE
PBK modeling is a critical computational method for extrapolating in vitro concentrations to in vivo doses.
- 1. Principle: A PBK model is a mathematical representation of the body as a series of compartments (e.g., liver, fat, rapidly perfused tissues, slowly perfused tissues) connected by the circulatory system. The model describes the absorption, distribution, metabolism, and excretion (ADME) of a chemical based on its physicochemical properties and human physiology.
- 2. Model Development Workflow:
Figure 2: Workflow for Developing a Physiologically Based Kinetic (PBK) Model for In Vitro to In Vivo Extrapolation (IVIVE). The process involves parameterization, model implementation, simulation for prediction, and critical sensitivity analysis.
- 3. IVIVE Calculation:
- Obtain a Bioactive Concentration (C~bio~) from a NAM, such as the concentration causing 10% activity (AC~10~) in a ToxTracker assay.
- Input this C~bio~ as a target concentration in a key tissue (e.g., liver) within the PBK model.
- The model is then run backwards (reverse dosimetry) to calculate the daily human external dose required to achieve this C~bio~ in the target tissue.
- This predicted dose is the Point of Departure (POD) for risk assessment. The Margin of Exposure (MoE) is calculated as: MoE = POD / Human Exposure Estimate. A sufficiently large MoE (e.g., > 100) indicates a low risk.
The Scientist's Toolkit: Essential Research Reagents and Solutions
Successful implementation of NGRA relies on a suite of sophisticated research tools and platforms. The table below catalogues key solutions cited in the literature.
Table 2: Key Research Reagent Solutions for NGRA Implementation
| Tool/Reagent | Type | Primary Function in NGRA | Example Use Case |
|---|---|---|---|
| ToxTracker Assay [107] | In Vitro Reporter Assay | Provides mechanistic insight into genotoxicity by activating specific GFP-tagged cellular stress pathways. | Discriminates between clastogens, aneugens, and non-genotoxic carcinogens in early compound screening. [107] |
| ReproTracker Assay [107] | Stem Cell-Based In Vitro Model | Assesses developmental toxicity by visualizing key processes in early embryogenesis using reporter stem cell lines. | Identifying potential teratogenic risks of drug candidates during preclinical development. [107] |
| Leadscope Model Applier [104] | In Silico Software Suite | Applies (Q)SAR models and expert alerts for toxicity prediction; includes databases for read-across and carcinogenicity assessment (e.g., CPCA for N-nitrosamines). | Predicting a compound's potential for acute oral toxicity or skin sensitization as part of a Tier 1 screening battery. [104] |
| Organ-on-a-Chip (e.g., Lung-Chip) [102] [105] | Advanced In Vitro System | Microfluidic devices containing human cells that emulate the structure and function of human organs; allows for exposure at the air-liquid interface (ALI). | More physiologically relevant assessment of inhalation toxicity for pharmaceuticals or industrial chemicals. [102] |
| Physiologically Based Kinetic (PBK) Models [102] [108] | Computational Model | Quantitatively describes the ADME of a chemical in the body, enabling in vitro-to-in vivo extrapolation (IVIVE) for risk quantification. | Converting an in vitro bioactivity concentration (e.g., AC~50~) into a human equivalent external dose for risk characterization. [102] |
| Adverse Outcome Pathway (AOP) Framework [105] [107] | Knowledge Organization Framework | Structures mechanistic toxicological knowledge from a Molecular Initiating Event (MIE) to an Adverse Outcome (AO) across levels of biological organization. | Supporting the use of Mechanistic Data (e.g., from ToxTracker) in IATA for regulatory safety assessments. [107] |
Current Challenges and Future Perspectives
Despite significant progress, the widespread adoption of NGRA faces several challenges. A primary hurdle is the need for standardization and validation of NAMs to build confidence for regulatory acceptance. [103] Efforts are ongoing to establish performance-based standards and demonstrate the reliability and relevance of these new methods. [106] Furthermore, the integration of diverse data streams from different NAMs into a coherent weight-of-evidence conclusion requires robust data integration strategies and the application of the FAIR principles (Findable, Accessible, Interoperable, and Reusable) to ensure data quality and utility. [106]
The future of NGRA is intrinsically linked to technological advancement. The increasing use of artificial intelligence (AI) and machine learning (ML) will enhance the predictive power of in silico models and improve data analysis. [105] [106] International initiatives, such as the European Partnership for the Assessment of Risks from Chemicals (PARC), are crucial for aligning research priorities and fostering the collaboration needed to qualify NAMs for regulatory use. [102] As these tools mature and the framework is refined, NGRA is poised to become the global standard for chemical safety assessment, enabling more human-relevant, efficient, and mechanistic-based protection of human health and the environment. [102] [109] [103]
Next Generation Risk Assessment (NGRA) represents a paradigm shift in toxicology, moving towards a more efficient, mechanistic, and hypothesis-driven framework for safety decisions. It leverages New Approach Methodologies (NAMs) to increase the efficiency of testing and reduce reliance on animal experiments [110]. This case study applies a tiered NGRA approach to pyrethroid insecticides, a class of chemicals critical for public health—particularly in controlling mosquito vectors of diseases like dengue and Zika—but which face the major challenge of widespread resistance [111]. The core of this NGRA is its exposure-driven and tiered structure, which allows for risk assessment with cost-effectiveness and limited use of vertebrate testing [110].
Background: Pyrethroids and the Resistance Crisis
Pyrethroids are a cornerstone of vector control, primarily targeting the voltage-gated sodium channels (VGSCs) in insect neurons. However, their efficacy is being severely undermined by resistance.
Key Genetic Resistance Mechanisms
The primary mechanism of resistance is knockdown resistance (kdr), caused by point mutations in the gene encoding the VGSC. These mutations reduce the insect's sensitivity to pyrethroids [111]. The table below summarizes major kdr mutations identified in Aedes aegypti populations.
Table 1: Functionally Confirmed kdr Mutations in Aedes aegypti [111]
| Mutation | Phenotypic Effect |
|---|---|
| V253F | Reduced pyrethroid binding to the sodium channel |
| V410L | Altered channel gating kinetics |
| L982W | Modified channel function and sensitivity |
| I1011M | Interference with insecticide action |
| V1016G | Decreased neuronal excitation |
| F1534C | Impaired insecticide binding |
Quantifying the Resistance Problem
The impact of these mechanisms is quantifiable through bioassays, revealing a severe resistance problem globally.
Table 2: Documented Pyrethroid Resistance Intensities in Aedes aegypti [111]
| Resistance Context | Pyrethroid | Resistance Ratio |
|---|---|---|
| Field-Selected Populations | Deltamethrin | Up to 249-fold |
| Field-Selected Populations | Permethrin | Exceeding 500-fold |
| Laboratory-Selected Strains | Multiple Pyrethroids | Can surpass 1,000-fold |
Tiered NGRA Framework for Pyrethroids
The proposed NGRA framework is structured in tiers of increasing specificity and complexity, making best use of existing information before initiating new tests [110]. The following diagram illustrates the workflow of this tiered strategy.
Tier 1: Prioritization and Initial Screening
The initial tier uses in silico tools and existing data for a high-throughput, low-cost screening of pyrethroids and their transformation products.
In Silico Exposure and Hazard Profiling
Quantitative Structure-Activity Relationship (QSAR) models are employed to predict key environmental fate parameters and ecotoxicological endpoints without new experimental data [1] [2]. These models use the chemical structure of a pesticide to forecast its behavior and effects.
Key Parameters for In Silico Modeling:
- Physical-Chemical Properties: Molecular weight, water solubility, vapor pressure, and the n-octanol-water partition coefficient (KOW) are fundamental for predicting environmental distribution [112].
- Environmental Fate: Using these properties, models can estimate a chemical's potential for runoff, leaching, and bioaccumulation. For instance, the Henry's Law Constant can be calculated from vapor pressure and water solubility to assess volatility [112].
- Toxicity Prediction: QSAR models can provide initial estimates of toxicity to non-target species, such as fish, Daphnia, and honeybees, helping to prioritize chemicals for further testing [2].
Application to Pyrethroid Resistance
At this tier, existing data on kdr mutation frequencies and resistance ratios from bioassays (like the examples in Table 2) can be used to prioritize pyrethroid formulations or identify geographical areas where resistance is most critical, guiding targeted interventions [111].
Tier 2: Mechanistic Investigation
If Tier 1 indicates potential risk or data gaps, the assessment proceeds to Tier 2, which utilizes more complex NAMs to understand the mechanisms of toxicity and resistance.
Experimental Protocol: Voltage-Gated Sodium Channel Binding Assay
This assay investigates the direct interaction between pyrethroids and their target site.
Objective: To quantify the differences in binding affinity of pyrethroids to wild-type and mutant (kdr) VGSCs. Methodology:
- Cell Line Preparation: Use a heterologous expression system (e.g., Xenopus oocytes or human HEK293 cells) to express wild-type or mutant insect VGSCs.
- Electrophysiology: Employ whole-cell voltage-clamp techniques to measure sodium currents.
- Pyrethroid Application: Apply increasing concentrations of the pyrethroid insecticide to the extracellular solution.
- Data Analysis: Quantify the compound's effect by measuring the percentage modification of the sodium current tail amplitude. Calculate the half-maximal effective concentration (EC50) for the wild-type and mutant channels. A higher EC50 in the mutant channel confirms reduced sensitivity due to the kdr mutation.
In Vitro Metabolism and Toxicokinetics
These studies assess how the insect metabolizes the pyrethroid, which is a major non-target resistance mechanism.
- Assay Systems: Utilize insect hepatocyte (or analogous) cell lines or sub-cellular fractions (e.g., microsomes) containing metabolic enzymes like cytochrome P450s.
- Analysis: Incubate the pyrethroid with the test system and use Liquid Chromatography-Mass Spectrometry (LC-MS/MS) to track the parent compound's depletion and the formation of metabolites over time. This provides data on metabolic stability and clearance rates.
Tier 3: Definitive Risk Characterization and Integrated Assessment
The final tier integrates exposure and effects data for a comprehensive risk characterization, using more complex models and targeted testing where necessary.
Toxicokinetic-Toxicodynamic (TK-TD) Modeling
TK-TD models move beyond simple concentration-response curves by describing the processes of uptake, distribution, metabolism, and elimination (TK) of a chemical, and the subsequent interactions with biological targets that lead to the toxic effect (TD). For pyrethroids, a TK-TD model can simulate how different kdr mutations or metabolic rates (TK) alter the probability of insect mortality (TD) under specific exposure scenarios [1].
Population-Level and Environmental Exposure Modeling
- Agent-Based Population Models: These models simulate the impact of pyrethroid resistance on mosquito population dynamics and disease transmission. They can incorporate genetic information (e.g., frequency of kdr alleles) to predict how mosquito populations recover after insecticide application.
- Environmental Exposure Models: Tools like the AGricultural DISPersal model (AGDISP) predict pesticide drift during application, helping to quantify off-target exposure for aquatic and terrestrial organisms [2]. For fate in water, models like TOXSWA simulate pesticide concentrations in ditches and streams, accounting for processes like adsorption to sediment and degradation [2].
The Scientist's Toolkit: Key Research Reagents and Solutions
Successful implementation of this NGRA framework relies on a suite of specialized reagents and tools.
Table 3: Essential Research Reagents and Solutions for Pyrethroid NGRA
| Category / Reagent | Function and Application in NGRA |
|---|---|
| Molecular Biology | |
| Cloned Insect VGSC Genes (Wild-type & kdr mutants) | Essential for heterologous expression in Tier 2 mechanistic studies to isolate and study the effect of specific mutations. |
| siRNA/shRNA for Resistance Genes | Used to knock down gene expression (e.g., specific P450s) in cell-based assays to confirm their role in metabolic resistance. |
| Cell-Based Assays | |
| Heterologous Expression Cell Lines (e.g., HEK293, Sf9) | Provide a controlled system for expressing target proteins (like VGSCs) for high-quality, reproducible Tier 2 screening. |
| Insect Hepatocyte or Cell Lines | Used for in vitro metabolism studies (Tier 2) to assess metabolic resistance and compound degradation. |
| Analytical Chemistry | |
| Analytical Standards (Pyrethroids & Metabolites) | Certified reference materials required for calibrating analytical instruments (e.g., LC-MS/MS) to accurately quantify concentrations in fate and metabolism studies. |
| In Silico Tools | |
| QSAR Software (e.g., EPI Suite) | Provides predictions for physical-chemical properties and environmental fate parameters in Tier 1 screening [112] [2]. |
| Toxicokinetic-Toxicodynamic (TK-TD) Modeling Platforms | Software used in Tier 3 to build mathematical models that link internal pesticide doses to dynamic biological effects over time [1]. |
The tiered NGRA approach for pyrethroid insecticides demonstrates a path forward for managing insecticide resistance and environmental impact in a more predictive, mechanistic, and efficient manner. By integrating in silico tools, in vitro bioassays, and sophisticated modeling, this framework aligns with the global push to reduce animal testing while improving the quality of risk assessment. For pyrethroids, this means explicitly accounting for real-world factors like kdr mutations, which is a critical step in developing resilient, evidence-based vector control interventions to mitigate the growing public health threat of arboviruses [111]. The success of NGRA ultimately depends on continued dialogue and collaboration to adapt legislative frameworks and regulatory guidance to accommodate these innovative methodologies [110].
In silico environmental risk assessment (ERA) represents a paradigm shift in how researchers and regulators evaluate the potential impact of chemicals, including pharmaceuticals and pesticides, on ecosystems. Defined as the use of computational methodologies to predict environmental fate and effects, this approach is gaining significant traction within modern regulatory frameworks. The drive towards New Approach Methodologies (NAMs) is fueled by the need to address complex challenges such as chemical mixture toxicity, cumulative exposure, and the environmental impact of transformation products, which are difficult to assess using traditional testing methods alone [113] [1]. This whitepaper provides a technical analysis of the current regulatory acceptance of in silico tools in the European Union (EU) and the United States (US), detailing the established pathways, persistent barriers, and future directions for their integration into environmental risk assessment.
Current Regulatory Frameworks and Acceptance
The European Union Landscape
The EU's regulatory framework for chemicals, including plant protection products (PPPs) and pharmaceuticals, provides a structured yet adaptable foundation for incorporating in silico methods.
Plant Protection Products (PPPs): The approval of active substances in PPPs is governed by Regulation (EC) No 1107/2009, which operates under a pre-marketing risk assessment principle [113] [114]. The foundational data requirements are outlined in Regulations (EU) No 283/2013 and 284/2013, which, while traditionally reliant on standardized experimental studies, are increasingly accommodating computational data. The International Uniform Chemical Information Database (IUCLID), developed by the European Chemicals Agency (ECHA) in collaboration with the OECD, is a critical platform for managing and submitting toxicological and ecotoxicological data in a harmonized format, facilitating the use of standardized data for risk assessment [113] [114].
Pharmaceuticals: For human medicinal products, the revised EMA guideline on environmental risk assessment (ERA), effective September 2024, mandates an ERA for all new marketing authorisation applications [115]. This guideline explicitly encourages a tiered, weight-of-evidence approach, where in silico models can play a vital role in filling data gaps, particularly in problem formulation and prioritization [115]. The guidance embeds the principles of the 3Rs (Replacement, Reduction, and Refinement), promoting the sharing of data to avoid unnecessary animal testing [115].
While not a formal acceptance criterion, the use of in silico tools is supported through guidelines from the European Food Safety Authority (EFSA) and the OECD. EFSA's scientific opinions often address cumulative exposure and mixture toxicity, areas where computational modeling is indispensable [113]. Furthermore, the EU's Green Deal and "Zero pollution" ambition are creating political impetus for more efficient and holistic assessment tools, indirectly bolstering the case for in silico methodologies [116].
The United States Landscape
In the US, the Environmental Protection Agency (USEPA) has long championed modern toxicity testing approaches for pesticide registration [117]. The agency collaborates with stakeholders to develop and implement innovative approaches that strengthen health and environmental protections.
However, the adoption of 21st-century testing approaches, including in silico methods, has been slower than anticipated. A significant barrier is the retention of outdated data requirements codified in the Code of Federal Regulations [117]. These prescriptive requirements can slow the uptake of New Approach Methodologies (NAMs) because the regulatory language is often tied to traditional animal testing protocols. The USEPA faces the dual challenge of needing sufficient resources to evaluate and establish confidence in new methods while navigating its existing regulatory architecture [117].
Comparative Analysis: EU vs. US
Table 1: Comparative Overview of In Silico ERA Regulatory Frameworks
| Aspect | European Union (EU) | United States (US EPA) |
|---|---|---|
| Primary Legislation | Regulation (EC) No 1107/2009 (PPPs); Directive 2001/83/EC (Pharmaceuticals) [113] [115] | Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA); Federal Food, Drug, and Cosmetic Act (FFDCA) [117] |
| Guidance for In Silico | Supported under EFSA/EMA guidelines, especially for data gaps and mixture assessment; explicit in revised 2024 Pharma ERA guide [113] [115] | Actively promotes modern testing but hampered by codified data requirements [117] |
| Key Driver for NAMs | European Green Deal; 3Rs policy; regulatory need to assess chemical mixtures [113] [116] | Efficiency goals; animal welfare concerns (reduction, refinement, replacement) [117] |
| Major Adoption Barrier | Need for validation and standardization across Member States; legal acceptance hurdles [113] | Outdated codified data requirements; resource constraints for method validation [117] |
| Data Management | IUCLID platform for standardized data submission and assessment [113] | Not specified in search results |
Critical Methodologies and Experimental Protocols
The application of in silico ERA relies on a suite of sophisticated computational tools. The following tiered approach is commonly adopted, moving from simpler, screening-level assessments to more complex, mechanistic modeling.
Tiered Approach and Problem Formulation
The first step in any ERA is problem formulation, which scopes the assessment based on data availability, time, and resources [1]. A tiered approach allows for assessment termination if early tiers indicate a low risk, saving resources for higher-risk scenarios.
Key In Silico Methodologies
Quantitative Structure-Activity Relationship (QSAR) Models
- Principle: QSAR models establish a quantitative relationship between the chemical structure of a compound (descriptors) and its biological activity or property (e.g., toxicity, biodegradability) [1].
- Protocol:
- Endpoint Selection: Define the property to be predicted (e.g., acute aquatic toxicity to fish, persistence (P)).
- Data Curation: Compile a high-quality dataset of chemical structures and their associated experimental endpoint values for a training set.
- Descriptor Calculation: Use software to compute molecular descriptors (e.g., logP, molecular weight, topological indices) for each compound.
- Model Building: Apply a statistical or machine learning algorithm (e.g., linear regression, partial least squares, random forest) to correlate descriptors with the endpoint.
- Validation: Assess model performance using internal (cross-validation) and external (test set) validation to ensure its predictive power and applicability domain are defined.
- Application: Used as a first-tier screening tool to predict ecotoxicity and environmental fate parameters for data-poor chemicals, or to prioritize chemicals for further testing [1] [24]. For example, QSAR models can be used to screen pharmaceuticals and their transformation products for potential carcinogenicity or mutagenicity [24].
Toxicokinetic-Toxicodynamic (TK-TD) and Dynamic Energy Budget (DEB) Models
- Principle: These biologically-based models move beyond simple endpoint prediction to simulate the processes that lead to toxicity over time. TK describes the uptake, distribution, metabolism, and excretion of a chemical in an organism, while TD describes the processes of damage and recovery at the target site. DEB models integrate these effects with the organism's energy allocation rules [1].
- Protocol:
- Model Parameterization: Gather data on species-specific life-history traits (e.g., growth, reproduction) from the literature and chemical-specific TK parameters (e.g., uptake and elimination rates) from experiments or QSARs.
- Model Calibration: Expose the model to existing toxicity data for a reference compound to calibrate the TD parameters.
- Simulation and Prediction: Run the model to simulate the effects of a chemical over the entire lifespan of an organism under various exposure scenarios, predicting sub-lethal effects like reduced growth or reproduction.
- Application: Used for a more realistic, mechanistically grounded risk assessment, particularly for chronic exposures and population-level effects [1].
Physiologically Based Pharmacokinetic (PBPK) Models
- Principle: A specific type of TK model that scales the distribution and metabolism of a chemical across different tissues and organs based on physiological parameters (e.g., blood flow, organ volume) [1].
- Application: While more common in human health risk assessment, they are increasingly applied in ecotoxicology for cross-species extrapolation.
Application to Complex Scenarios: Chemical Mixtures and Transformation Products
A major strength of in silico approaches is their ability to address the "cocktail effect" of chemical mixtures, a realistic exposure scenario confirmed by monitoring data showing multiple pesticide residues in most food samples [113] [114]. Computational models can predict interactions (additive, synergistic, antagonistic) based on the modes of action of the individual components [113]. Furthermore, in silico tools are invaluable for assessing transformation products (TPs), which are formed when parent compounds degrade in the environment or during water treatment. Since TPs are often unknown and lack analytical standards, in silico methods may be the only viable option for an initial hazard assessment [24].
The Scientist's Toolkit: Essential Research Reagents and Solutions
Table 2: Key Research Reagents and Computational Tools for In Silico ERA
| Tool Category | Specific Examples | Function & Application |
|---|---|---|
| QSAR Software & Platforms | ECOSAR, VEGA Platform, OECD QSAR Toolbox | Predicts ecotoxicological endpoints and environmental fate properties from molecular structure. Used for screening and priority setting [1]. |
| Toxicology Databases | EPA's CompTox Chemicals Dashboard, ECHA database | Provides curated experimental data (e.g., toxicity, physicochemical) essential for model training and validation [1]. |
| Read-Across Frameworks | As part of REACH and OECD guidance | Fills data gaps for a "target" chemical by using data from similar "source" chemicals (based on structure, properties, mode of action) [1]. |
| TK-TD & DEB Modeling Platforms | DEBtool, GUTS (General Unified Threshold model of Survival) | Provides a mathematical framework to simulate the effects of chemicals on organisms over time, under dynamic exposure conditions [1]. |
| Chemical Structure Drawing & Descriptor Software | ChemDraw, RDKit, PaDEL-Descriptor | Used to draw chemical structures and calculate molecular descriptors required as input for QSAR and other property prediction models [1]. |
The future of in silico ERA is promising but requires concerted effort across multiple fronts to achieve full regulatory maturity.
- Method Validation and Standardization: Establishing universally accepted protocols for validating in silico models is paramount. This includes defining applicability domains and performance metrics to build regulatory and scientific confidence [113] [117].
- Regulatory Modernization: As highlighted in the US, a shift from prescriptive, checklist-based data requirements to a more flexible, knowledge-based framework is essential to facilitate the uptake of NAMs [117].
- Addressing Complex Systems: Future development must focus on integrating in silico models to tackle multi-stressor environments. This includes linking chemical fate models with ecological models to assess impacts on biodiversity and ecosystem services, aligning with the EU's push for a systems-based environmental risk assessment [1] [116].
- Open Data and Collaboration: Enhancing data sharing through platforms like IUCLID and promoting the use of publicly available data, as encouraged in the revised EMA guideline, will enrich the data pool for model development and testing [113] [115].
In conclusion, while the EU and US regulatory frameworks are progressively embracing in silico methodologies, the journey towards their full integration is ongoing. The EU demonstrates a structured, if sometimes cautious, approach through specific guidelines and databases, whereas the US faces distinct challenges rooted in its established regulatory code. For researchers and regulators, the path forward lies in continued collaboration to refine these powerful tools, validate their predictive capabilities, and modernize regulatory frameworks to enable a more efficient, protective, and holistic assessment of environmental risks.
Conclusion
In silico environmental risk assessment represents a paradigm shift towards a more efficient, ethical, and mechanistic understanding of chemical safety. By leveraging computational models, researchers can proactively identify hazards, fill critical data gaps, and assess risks for a vast number of chemicals, including those in development. The integration of these tools within frameworks like AOPs and tiered NGRA allows for a more nuanced assessment of complex scenarios, such as combined exposures to multiple chemicals. For the biomedical and clinical research community, the adoption of in silico ERA promises to accelerate drug development by enabling early de-risking of compounds and their potential environmental metabolites. Future progress hinges on expanding robust databases, refining models for transformation products, and fostering wider regulatory harmonization, ultimately paving the way for a truly animal-free, predictive toxicology future.
References
- 1. In Silico Methods for Environmental Risk Assessment [https://pubmed.ncbi.nlm.nih.gov/35188648/]
- 2. In silico environmental risk assessment improves efficiency ... [https://www.sciencedirect.com/science/article/abs/pii/S0048969723065051]
- 3. Unlocking the potential of in silico chemical safety ... [https://www.sciencedirect.com/science/article/pii/S2468111318301397]
- 4. CAse studies on the Development and Application of in-Silico ... [https://cordis.europa.eu/project/id/212668/reporting/fr]
- 5. In Silico Hazard Screening and Risk Evaluation [https://pmc.ncbi.nlm.nih.gov/articles/PMC11768813/]
- 6. In-silico chemical evaluation at CEA | CEA News [https://cea.adas.co.uk/news/in-silico-chem-evaluation-at-cea/]
- 7. Framework for risk assessment of PFAS utilizing ... [https://pubmed.ncbi.nlm.nih.gov/35026182/]
- 8. An in Silico Environmental Risk Assessment of Palm Oil ... [https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5265532]
- 9. A combination of high-throughput in vitro and in silico new ... [https://academic.oup.com/etc/article/44/9/2599/7942992]
- 10. In silico toxicology protocols - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6026539/]
- 11. The use of new approach methodologies for ... [https://www.sciencedirect.com/science/article/abs/pii/S2468584422000915]
- 12. Comparison of in silico, in vitro, and in vivo toxicity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11217893/]
- 13. In Silico Methods for Hazard Characterization [https://www.intertek.com/assuris/safety/in-silico-methods-for-hazard-characterization/]
- 14. ( Adverse ) development I: strategies and... outcome pathway AOP [https://pubmed.ncbi.nlm.nih.gov/25466378/]
- 15. Eawag - Swiss Federal Institute of Aquatic Science and Technology... [https://www.eawag.ch/en/department/utox/projects/adverse-outcome-pathways/]
- 16. From Qualitative to Quantitative AOP: A Case Study of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9006165/]
- 17. Towards a qAOP framework for predictive toxicology [https://www.sciencedirect.com/science/article/pii/S2468111321000414]
- 18. Limitations of Animal Studies for Predicting Toxicity in Clinical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6978558/]
- 19. Frontiers | Advancing human health risk : the role of new... assessment [https://www.frontiersin.org/journals/toxicology/articles/10.3389/ftox.2025.1632941/full]
- 20. A review on in prediction of the silico posed by... environmental risks [https://pubmed.ncbi.nlm.nih.gov/38008816/]
- 21. QSAR Toolbox [https://qsartoolbox.org/]
- 22. in Silico Tools [https://www.vegahub.eu/portfolio-types/in-silico-tools/]
- 23. A Framework that Considers the Impacts of Time, Cost, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9290960/]
- 24. of fate and effects of... In silico environmental risk assessment [https://pubs.rsc.org/en/content/articlelanding/2023/ew/d2ew00518b]
- 25. From animal testing to in Silico models: a systematic review ... [https://link.springer.com/article/10.1007/s10260-025-00794-0]
- 26. Highlighting best practices to advance next-generation risk ... [https://www.sciencedirect.com/science/article/pii/S3050620425000156]
- 27. ECHA Releases 2025 Edition of "Key Areas of Regulatory ... [https://www.cirs-group.com/en/chemicals/echa-releases-2025-edition-of-key-areas-of-regulatory-challenge-report]
- 28. Next generation risk assessment and new approach ... [https://www.sciencedirect.com/science/article/pii/S0300483X25001702]
- 29. REACH Regulation - Environment - European Commission [https://environment.ec.europa.eu/topics/chemicals/reach-regulation_en]
- 30. The Importance of Toxicological Assessment for Beauty ... [https://www.alsglobal.com/en/News-and-publications/2025/08/cosmetics-safety-toxicological-dossier]
- 31. Understanding EU Cosmetic Regulation (EC 1223/2009) [https://www.schlafenderhase.com/shblog/eu-cosmetic-regulation]
- 32. Cosmetic Industry Regulations 2025: FDA Compliance Guide [https://elchemy.com/blogs/personal-care/cosmetic-industry-regulations-in-2025-what-brands-must-know-about-compliance-and-the-fda-cosmetics-act]
- 33. In Silico Frontiers Shaping the Next Generation of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12445007/]
- 34. Use maps - ECHA - European Union [https://echa.europa.eu/csr-es-roadmap/use-maps/use-maps-library]
- 35. QSAR Modeling: Where have you been? Where are you going ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4074254/]
- 36. Beyond QSARs: Quantitative Knowledge–Activity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12646586/]
- 37. Comparative Study of (Q)SAR Models for Predicting the ... [https://www.sciencedirect.com/science/article/pii/S3050620425000491]
- 38. CheS-Mapper 2.0 for visual validation of (Q)SAR models [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-014-0041-7]
- 39. In Silico Methods for Environmental Risk Assessment [https://link.springer.com/protocol/10.1007/978-1-0716-1960-5_23]
- 40. Advancing the Science of a Read-across Framework for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11880891/]
- 41. Read-Across: A Practical New Approach Methodology for ... [https://www.intertek.com/blog/2025/07-23-read-across/]
- 42. Principles, Techniques and Frameworks - Risk Assessment [https://assessments.epa.gov/risk/document/&deid%3D355368]
- 43. Current trends in read-across applications for chemical risk ... [https://pubmed.ncbi.nlm.nih.gov/36250612/]
- 44. Next-generation risk assessment read-across case study [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2024.1421601/full]
- 45. Integrating in silico models and read-across methods for ... [https://www.sciencedirect.com/science/article/pii/S0160412019312681]
- 46. Advancing human health risk assessment: the role of new ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12537679/]
- 47. Recommendations to address uncertainties in ... [https://www.nature.com/articles/s41598-019-47698-0]
- 48. Review Dynamic energy budget models in ecological risk ... [https://www.sciencedirect.com/science/article/pii/S0048969718304455]
- 49. Environmental Risk Assessment with Energy Budget Models ... [https://academic.oup.com/etc/article/43/2/440/7728729]
- 50. Incorporating Suborganismal Processes into Dynamic Energy ... [https://repository.library.noaa.gov/view/noaa/62456]
- 51. Environmental Risk Assessment of Time-Variable Toxicant ... [https://pubmed.ncbi.nlm.nih.gov/39221922]
- 52. Toxicokinetic-toxicodynamic modeling reveals the ... [https://www.sciencedirect.com/science/article/abs/pii/S0269749125011650]
- 53. Environmental Risk Assessment with Energy Budget Models [https://pubmed.ncbi.nlm.nih.gov/38051527/]
- 54. Using Predictive Methods to Assess Exposure and Fate ... [https://www.epa.gov/tsca-screening-tools/using-predictive-methods-assess-exposure-and-fate-under-tsca]
- 55. What are Predictive Models and Tools under TSCA? [https://www.epa.gov/tsca-screening-tools]
- 56. Insights from modeling the fate of and exposure to persistent ... [https://academic.oup.com/etc/article-abstract/44/11/3349/8205747]
- 57. Mapping the Rise in Machine Learning in Environmental ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12567794/]
- 58. Exposure Modeling [https://waterborne-env.com/exposure-modeling/]
- 59. In silico Safety and Toxicology assessment: ToxStudio [https://www.certara.com/webinar/in-silico-safety-and-toxicology-assessment-toxstudio/]
- 60. Coupled In Silico Toxicology Models Reveal Equivalent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12390238/]
- 61. In silico methods for immunogenicity risk assessment and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10978032/]
- 62. Closing data gaps on the hazards of BPA alternatives for ... [https://www.eu-parc.eu/projects/closing-data-gaps-hazards-bpa-alternatives-human-health]
- 63. Adverse Outcome Pathway (AOP) Research Brief [https://www.epa.gov/chemical-research/adverse-outcome-pathway-aop-research-brief]
- 64. The Adverse Outcome Pathway: A Multifaceted Framework ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5906804/]
- 65. Challenges and Opportunities for Integrating In Silico ... [https://www.mdpi.com/2073-4441/12/12/3549]
- 66. Validation of a blood biomarker panel for machine learning ... [https://www.nature.com/articles/s41598-024-74953-w]
- 67. Clinical Validation of a Machine Learning-Based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11857289/]
- 68. Machine learning framework to extract the biomarker ... [https://www.sciencedirect.com/science/article/pii/S2001037024000618]
- 69. AI and the future of biomarker analysis in early R&D [https://www.drugtargetreview.com/article/190428/ai-and-the-future-of-biomarker-analysis-in-early-rd/]
- 70. The use of new approach methodologies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9890323/]
- 71. The VEGA Tool to Check the Applicability Domain Gives ... [https://www.mdpi.com/1422-0067/24/12/9894]
- 72. Assessing risk of bias in toxicological studies in the era ... [https://link.springer.com/article/10.1007/s00204-025-03978-5]
- 73. Common issues of data science on the eco-environmental ... [https://www.sciencedirect.com/science/article/pii/S0160412025000522]
- 74. A framework for categorizing sources of uncertainty in ... [https://www.sciencedirect.com/science/article/pii/S0273230024001788]
- 75. In Silico Frontiers Shaping the Next Generation of ... [https://pubmed.ncbi.nlm.nih.gov/40892019/]
- 76. A continuous in silico learning strategy to identify safety ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10025185/]
- 77. A new integrated in silico strategy for the assessment and ... [https://www.sciencedirect.com/science/article/abs/pii/S0160412015301240]
- 78. In silico methods for drug-target interaction prediction - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12570315/]
- 79. A Guide to In Silico Drug Design - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC9867171/]
- 80. The Transformative Power of In Silico Experiments ... [https://www.chemcopilot.com/blog/silico-experiments-modern-research-laboratory]
- 81. Developing water quality criteria and assessing ecological ... [https://www.sciencedirect.com/science/article/abs/pii/S026974912501677X]
- 82. Coupled In Silico Toxicology Models Reveal Equivalent ... [https://www.mdpi.com/2305-6304/13/8/671]
- 83. ICE: Integrated Chemical Environment [https://ntp.niehs.nih.gov/whatwestudy/niceatm/comptox/ct-ice/ice]
- 84. Application of Interspecies Correlation Estimation (ICE) ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7848808/]
- 85. Coupled In Silico Toxicology Models Reveal Equivalent ... [https://pubmed.ncbi.nlm.nih.gov/40863947/]
- 86. ICE tools to support chemical evaluations [https://www.sciencedirect.com/science/article/pii/S2468111321000323]
- 87. NAM Use for Regulatory Application - SARA-ICE [https://www.pcrm.org/ethical-science/animal-testing-and-alternatives/nura/sara-ice]
- 88. A problem formulation framework for the application of in silico ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11106140/]
- 89. A Statistical Exploration of QSAR Models in Cancer Risk ... [https://www.mdpi.com/2305-6304/13/4/299]
- 90. A tiered next-generation risk assessment framework ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12198285/]
- 91. The methodology of quantitative risk assessment studies [https://ehjournal.biomedcentral.com/articles/10.1186/s12940-023-01039-x]
- 92. Quantifying the benefits of using read-across and in silico ... [https://www.sciencedirect.com/science/article/pii/S0273230016302525]
- 93. In Silico Research Is Rewriting the Rules of Drug Development [https://pmc.ncbi.nlm.nih.gov/articles/PMC12070237/]
- 94. In silico trials: Verification, validation and uncertainty ... [https://www.sciencedirect.com/science/article/pii/S1046202319302452]
- 95. Applicability of In Silico New Approach Methods for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12087733/]
- 96. How in silico and QSAR approaches can increase confidence in environmental hazard and risk assessment - PubMed [https://pubmed.ncbi.nlm.nih.gov/30447098/]
- 97. Environmental Risk Assessment of Pharmaceuticals [https://www.zeclinics.com/blog/environmental-risk-assessment-of-pharmaceuticals]
- 98. ERA (Environmental Risk Assessment) of Medicinal Products [https://toxhub-consulting.com/2025/06/18/era/]
- 99. TOOLS In silico off-target profiling for enhanced drug safety ... [https://www.sciencedirect.com/science/article/pii/S2211383524000844]
- 100. New approach methodologies (NAMs): identifying and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10964841/]
- 101. The IHI VICT3R project for reducing the use of animals by ... [https://www.sciencedirect.com/science/article/pii/S3050620425000600]
- 102. 综述:新一代风险评估和新方法学在安全可持续设计化学品与 ... [https://www.ebiotrade.com/newsf//2025-6/20250618171139985.htm]
- 103. 基于新途径方法的下一代风险评估研究进展 [https://ccj.pku.edu.cn/article/info?id=383670641]
- 104. Leadscope Model Applier 2025.0 - 增强预测毒理学的新功能 [https://www.instem.com/zh/leadscope-model-applier-version-2025-0-new-features-for-enhanced-predictive-toxicology/]
- 105. 2023(第六届)毒性测试替代方法与转化毒理学(国际)学术 ... [https://chntox.org/home/xshd/xshd_detail/id/56.html]
- 106. 综述:推进人类健康风险评估:新方法学(NAM)的作用 [https://www.ebiotrade.com/newsf/2025-10/20251008000957812.htm]
- 107. 机制毒理检测领域的专家 [https://toxys.com/toxys-china/]
- 108. 云南省疾病预防控制中心 [https://www.yncdc.cn/dtxx/gjgn/2025/15233.shtml]
- 109. 环境健康风险评估制度建设的国际经验与中国路径 [https://www.hjwsxzz.com/article/doi/10.13421/j.cnki.hjwsxzz.2025.10.002?viewType=HTML]
- 110. Next Generation Risk Assessment approaches for ... [https://www.sciencedirect.com/science/article/pii/S2452074824000338]
- 111. Pyrethroid resistance in Aedes aegypti: genetic mechanisms ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12522378/]
- 112. Guidance for Reporting on the Environmental Fate and ... [https://www.epa.gov/pesticide-science-and-assessing-pesticide-risks/guidance-reporting-environmental-fate-and-transport]
- 113. Scientific and Regulatory Perspectives on Chemical Risk Assessment of Pesticides in the European Union [https://www.mdpi.com/2039-4713/15/5/173]
- 114. Scientific and Regulatory Perspectives on Chemical Risk ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12565433/]
- 115. EU Requirements for Environmental Risk Assessment (ERA) [https://www.dlrcgroup.com/eu-requirements-for-environmental-risk-assessment-era/]
- 116. Improving the environmental risk assessment for plant ... [https://www.eu-parc.eu/projects/improving-environmental-risk-assessment-plant-protection-products]
- 117. Enhancing pesticide risk assessment processes at the US ... [https://www.sciencedirect.com/science/article/abs/pii/S0273230025001916]